ポリ(アクリル酸メチル),トルエン溶液 化学特性,用途語,生産方法
定義
本品は、アクリル酸メチルの重合体であり、次の化学式で表される。
解説
ポリアクリル酸メチル略称PMA.アクリル酸メチルを溶液重合,または乳化重合で得られる無色の無定形固体.ベンゼン,アセトン,酢酸エチルなどに可溶,エーテルには膨潤し,四塩化炭素や飽和炭化水素,アルコール類などに不溶.軟化点75 ℃,ガラス転移点3 ℃.300 ℃ 以上で分解する.アルカリによって加水分解する.ヒドラジンと反応し,種々の誘導体の原料になるほか,接着剤,塗料用に広く用いられる.繊維加工方面では補強,織物のり剤,不織布,バインダー用として利用され,紙加工や,合成ゴム,皮革加工にも用いられる.
化粧品の成分用途
皮膜形成剤
化学的特性
clear liquid
使用
PMA/methylamine borane (MeAB) composites, prepared by solution blending process finds uses as a hydrogen storage material with better dehydrogenation property compared to MeAB.
製造方法
The structure of methyl acrylate is H2C=CH-COOCH3. The monomer used to prepare poly(methyl acrylate) is produced by the oxidation of propylene. The resin is made by free-radical polymerization initiated by peroxide catalysts and has the following formula:
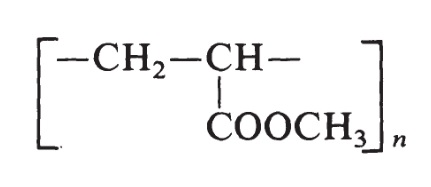
Poly(methyl acrylate) resins vary from soft, elastic, film-forming materials to hard plastics.
定義
ChEBI: An acrylate macromolecule composed of repeating methoxycarbonylethylene units.
調製方法
Prepared by the polymerization of acrylic and methacrylic acids or
their esters, e.g. butyl ester or dimethylaminoethyl ester
安全性
Polymethacrylate copolymers are widely used as film-coating
materials in oral pharmaceutical formulations. They are also used
in topical formulations and are generally regarded as nontoxic and
nonirritant materials.
Based on relevant chronic oral toxicity studies in rats and
conventionally calculated with a safety factor of 100, a daily intake
of 2–200 mg/kg body-weight depending on the grade of Eudragit
may be regarded as essentially safe in humans.
貯蔵
Dry powder polymer forms are stable at temperatures less than
30°C. Above this temperature, powders tend to form clumps,
although this does not affect the quality of the substance and the
clumps can be readily broken up. Dry powders are stable for at least
3 years if stored in a tightly closed container at less than 30°C.
Dispersions are sensitive to extreme temperatures and phase
separation occurs below 0°C. Dispersions should therefore be
stored at temperatures between 5 and 25°C and are stable for at
least 18 months after shipping from the manufacturer’s warehouse
if stored in a tightly closed container at the above conditions.
純化方法
Precipitate it from a 2% solution in acetone by addition of water.
不和合性
Incompatibilities occur with certain polymethacrylate dispersions
depending upon the ionic and physical properties of the polymer
and solvent. For example, coagulation may be caused by soluble
electrolytes, pH changes, some organic solvents, and extremes of
temperature. For example, dispersions of Eudragit L 30
D, RL 30 D, L 100-55, and RS 30 D are incompatible with
magnesium stearate. Eastacryl 30 D, Kollicoat MAE 100 P, and
Kollicoat MAE 30 DP are also incompatible with magnesium
stearate.
Interactions between polymethacrylates and some drugs can
occur, although solid polymethacrylates and organic solutions are
generally more compatible than aqueous dispersions.
規制状況(Regulatory Status)
Included in the FDA Inactive Ingredients Database (oral capsules
and tablets). Included in nonparenteral medicines licensed in the
UK. Included in the Canadian List of Acceptable Non-medicinal
Ingredients.
ポリ(アクリル酸メチル),トルエン溶液 上流と下流の製品情報
原材料
準備製品