| | 7446-09-5 Basic information More.. |
| Product Name: | Sulfur dioxide | | Synonyms: | sulfurous acid gas;Sulfur dioxide;Sulfur dioxide Standard;siarkidwutlenek(polish);Sulfur oxide (SO2);sulfur dioxide pressure tin with 100 ml (net ~128 G);SULFUR DIOXIDE, PRESSURE TIN WITH 100 ml (NET CA. 128 gm);SULFUR DIOXIDE, 99.9+% | | CAS: | 7446-09-5 | | MF: | O2S | | MW: | 64.06 | | EINECS: | 231-195-2 | | Mol File: | 7446-09-5.mol | 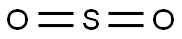 |
Use
Containers may explode in heat of fire or they may rupture and release irritating toxic Sulfur dioxide. Sulfur dioxide has explosive properties when Sulfur dioxide comes in contact with sodium hydride; potassium chlorate at elevated temperatures; ethanol; ether; zinc ethylsulfurinate at very cool temperatures (-15C); fluorine; chlorine trifluoride and chlorates. Sulfur dioxide will react with water or steam to produce toxic and corrosive fumes. When the liquid is heated Sulfur dioxide may release irritating, toxic Sulfur dioxide gas. Avoid ammonia, monocesium or monopotassium acetylide; dicesium monoxide; iron (II) oxide; tin oxide; lead (IV) oxide; chromium; manganese; molten sodium, powder aluminum and rubidium. Sulfur dioxide has explosive properties when Sulfur dioxide comes in contact with sodium hydride; potassium chlorate at elevated temperatures; ethanol; ether; zinc ethylsulfurinate at very cool temperatures (-15C); fluorine; chlorine trifluoride and chlorates. Sulfur dioxide will react with water or steam to produce toxic and corrosive fumes. Hazardous polymerization may not occur.
|
|
7446-09-5
Recommend Suppliers |
|