| | 7782-77-6 Basic information More.. |
| Product Name: | NITROUS ACID | | Synonyms: | Kyselina dusite;kyselinadusite;Nitrosyl hydroxide;Nitrous acid, trans;NITROUS ACID;HNO2;HONO;nitrousaci | | CAS: | 7782-77-6 | | MF: | HNO2 | | MW: | 47.01 | | EINECS: | 231-963-7 | | Mol File: | 7782-77-6.mol | 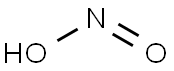 |
Use
Nitrous acid is unstable. It decomposes to form nitric acid and nitric oxide:
3HNO2 NO3 + H3O+ + 2NO
Strong oxidizing agents, such as permanganate, readily oxidize nitrous acid to nitric acid.
Nitrous acid is an effective oxidizing agent. It oxidizes hydrogen sulfide to sulfur forming either nitric oxide or ammonia, depending on the acidity of the solution:
2HNO2 + H2S S + 2NO + 2H2O
HNO2 + 3H2S 3S + NH3 + 2H2O
In acid medium it oxidizes iodide ion to iodine:
HNO2 + I + 6H+ 3I2 + NH3 + 2H2O
|
|
7782-77-6
Recommend Suppliers |
|