- Diquat ion
-

- $15.00 / 1KG
-
2021-08-12
- CAS:2764-72-9
- Min. Order: 1KG
- Purity: 99%+ HPLC
- Supply Ability: Monthly supply of 1 ton
- Diquat ion
-

- $15.00 / 1Kg/Bag
-
2020-11-16
- CAS:2764-72-9
- Min. Order: 1Kg/Bag
- Purity: 99%
- Supply Ability: 20 Tons
|
| | Diquat ion Basic information |
| Product Name: | Diquat ion | | Synonyms: | 6,7-Dihydro-5,8-didehydrodipyrido[1,2-a:2',1'-c]pyrazine-5,8-diium;6,7-Dihydrodipyrido[1,2-a:2',1'-c]pyrazine-5,8-diium;9,10-dihydro-8a,10a-diazoniaphenanthrene ion,diquat;D004178;Dibromide, diquat;1,1'-ethylene-2,2'-bipyridylium;6,7-dihydrodipyrido[1,2-a:2',1'-c]pyrazinediium;9,10-Dihydro-8a,10a-diazoniaphenanthrene | | CAS: | 2764-72-9 | | MF: | C12H12N2+2 | | MW: | 184.24 | | EINECS: | 220-433-0 | | Product Categories: | | | Mol File: | 2764-72-9.mol | 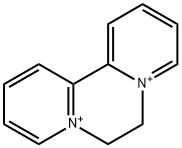 |
| | Diquat ion Chemical Properties |
| | Diquat ion Usage And Synthesis |
| Description | Diquat forms a monohydrate which consists ofcolorless to yellow crystals. Dibromide salt (herbicide) is ayellow crystal. The commercial product may be found in aliquid concentrate or a solution. Molecular weight= 344.08;Boiling point = not determined (decomposes); Freezing/Melting point = 335℃; Vapor pressure= 0.0001 mmHg at20℃. Hazard Identification (based on NFPA-704 M RatingSystem): Health 3, Flammability 1, Reactivity 0. Soluble inwater. | | Production Methods | Diquat is manufactured in the United Kingdom by the
oxidative coupling of two molecules of pyridine over a
heated Raney nickel catalyst to produce 2,20-bipyridyl,
which is reacted with ethylene dibromide in water to give
diquat dibromide. It is formulated worldwide in various
water-soluble products and in various concentrations, typically
2 lb/gal. | | Definition | ChEBI: The organic cation formed formally by addition of an ethylene bridge between the nitrogen atoms of 2,2'-bipyridine. Most often available as the dibromide. | | General Description | Diquat ion is a yellow crystalline solid dissolved in a liquid carrier. Diquat ion is a water emulsifiable liquid. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Since Diquat ion is a liquid Diquat ion can easily penetrate the soil and contaminate groundwater and nearby streams. Diquat ion can cause illness by inhalation, skin absorption and/or ingestion. Diquat ion is used as a herbicide. | | Air & Water Reactions | Water soluble. Solutions are stable in neutral and acid solutions; however, they are unstable in alkaline solutions. | | Reactivity Profile | Diquat ion is light sensitive. Diquat ion can corrode aluminum and other metals. | | Health Hazard | INHALATION: No appreciable vapor pressure. Prolonged contact with spray or mist may cause oral and nasal irritation. EYES: Irritation. SKIN: Irritation. INGESTION: Vomiting, diarrhea, general malaise. Possible kidney and liver damage, dyspnea, and pulmonary edema. With large doses there may be tremors or convulsions. OTHER: May be fatal if swallowed, inhaled, or absorbed through skin. | | Fire Hazard | Behavior in Fire: Decomposes at high temperature, charring rather than melting or boiling. | | Agricultural Uses | Herbicide, Desiccant: Diquat or diquat dibromide is a nonselective,
quick-acting herbicide and plant growth regulator, causing
injury only to the parts of the plant to which it is applied.
Diquat is referred to as a desiccant because it causes a leaf
or an entire plant to dry out quickly. It is used to desiccate
potato vines and seed crops, to control flowering of sugarcane, and for industrial and aquatic weed control in environments such as catfish farms. It is not residual; it does not
leave any trace of herbicide on or in plants, soil, or water. | | Trade name | AQUACIDE®; AQUA-CLEAR®[C]
AQUAKILL®; CLEANSWEEP®; DEIQUAT®;
DEXTRONE®; FARMON PDQ®; FB/2®; FEGLOX®;
GROUNDHOG SOLTAIR®; ORTHO DIQUAT®;
PATHCLEAR®; PREEGLONE®; REGLON®;
REGLONE®; Zeneca Ag Products; REGLOX®;
REWARD®; TAG®; TORPEDO®; VEGATROLE®;
WEEDOL (ICI)®; WEEDTRINE-D® | | Potential Exposure | AgriculturalChemical; Mutagen; Reproductive Effector; Human Data;Primary Irritant. Those involved in the manufacture, formulation, and application of this herbicide used to control awide range of aquatic and terrestrial weeds. | | First aid | If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit | | Carcinogenicity | Several carcinogenicity studies
have been conducted on diquat by the oral route.
In a 2-year feeding study at doses as high as 75 mg/kg in
mice, and a drinking water study at doses as high as
2.6 mg/L of water in rats, no evidence of tumorigenicity
was seen. Similar negative results were reported for diquat
in a 2-year feeding study (Hayes, 1991) in rats at dose levels
up to 720 mg/kg and in a 2-year drinking water study in mice
at doses of 2–4 mg/kg. | | storage | Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with diquatyou should be trained on its proper handling and storage.Store in tightly closed containers in a cool, well-ventilatedarea away from oxidizers and reducing agents. Where possible, automatically pump liquid from drums or other storagecontainers to process containers. Drums must be equippedwith self-closing valves, pressure vacuum bungs, and flamearresters. Use only nonsparking tools and equipment, especially when opening and closing containers of this chemical. Sources of ignition, such as smoking and open flames,are prohibited where this chemical is used, handled, orstored in a manner that could create a potential fire orexplosion hazard. Wherever this chemical is used, handled,manufactured, or stored, use explosion-proof electricalequipment and fittings. | | Shipping | Diquat dibromide requires a Corrosive liquid,n.o.s. label. It falls in Hazard Class 8 and Packing Group III. | | Incompatibilities | Keep away from alkalis, UV light, basicsolutions. Concentrated solutions of Diquat attackaluminum. |
| | Diquat ion Preparation Products And Raw materials |
|