|
|
| | 2β,14β-Ethano-15,20-didehydro-15,20-dideethyl-2,7α-dihydro-11-methoxycorynan-14-acetic acid methyl ester Basic information |
| Product Name: | 2β,14β-Ethano-15,20-didehydro-15,20-dideethyl-2,7α-dihydro-11-methoxycorynan-14-acetic acid methyl ester | | Synonyms: | 2β,14β-Ethano-15,20-didehydro-15,20-dideethyl-2,7α-dihydro-11-methoxycorynan-14-acetic acid methyl ester;Schizophyllin;Schizophylline;3H-Cyclopent[ij]indolo[2,3-a]quinolizine-13a(13bH)-acetic acid, 5,6,6a,11,12,13-hexahydro-9-methoxy-, methyl ester, (6aR,11aR,13aS,13bS)- | | CAS: | 2447-60-1 | | MF: | C21H26N2O3 | | MW: | 354.44 | | EINECS: | | | Product Categories: | | | Mol File: | 2447-60-1.mol | 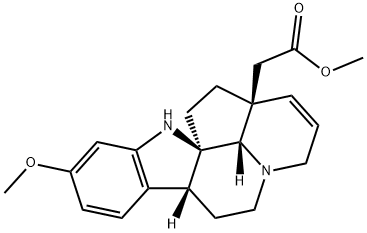 |
| | 2β,14β-Ethano-15,20-didehydro-15,20-dideethyl-2,7α-dihydro-11-methoxycorynan-14-acetic acid methyl ester Chemical Properties |
| | 2β,14β-Ethano-15,20-didehydro-15,20-dideethyl-2,7α-dihydro-11-methoxycorynan-14-acetic acid methyl ester Usage And Synthesis |
| Description | The roots and bark of Schizozygia caffaeoides (Boj.) Baili. also furnish this alkaloid which has the structure given above. It is 1aevorotatory with [α]24D - 64° (c 1.0, CHCI3) and gives an ultraviolet spectrum consisting of two absorption maxima at 251 and 299 mil. | | References | Renner, Kernweisz., Experientia, 19,244 (1963)
Structure:
Renner., Lloydia, 27,406 (1964) |
| | 2β,14β-Ethano-15,20-didehydro-15,20-dideethyl-2,7α-dihydro-11-methoxycorynan-14-acetic acid methyl ester Preparation Products And Raw materials |
|