|
|
| | (20S)-3β-(Dimethylamino)-4,4,14-trimethyl-20-(methylamino)-B(9a)-homo-19-nor-5α-pregna-9(11),9a-dien-16α-ol Basic information |
| Product Name: | (20S)-3β-(Dimethylamino)-4,4,14-trimethyl-20-(methylamino)-B(9a)-homo-19-nor-5α-pregna-9(11),9a-dien-16α-ol | | Synonyms: | (20S)-3β-(Dimethylamino)-4,4,14-trimethyl-20-(methylamino)-B(9a)-homo-19-nor-5α-pregna-9(11),9a-dien-16α-ol;Buxaminol B;Benzo[4,5]cyclohept[1,2-e]inden-2-ol, 9-(dimethylamino)-1,2,3,3a,4,7,8,9,10,10a,11,12,12a,12b-tetradecahydro-3a,10,10,12b-tetramethyl-3-[(1S)-1-(methylamino)ethyl]-, (2R,3S,3aR,9S,10aR,12aR,12bS)- | | CAS: | 58672-77-8 | | MF: | C27H46N2O | | MW: | 414.67 | | EINECS: | | | Product Categories: | | | Mol File: | 58672-77-8.mol | 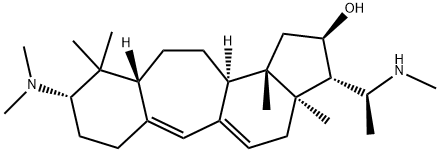 |
| | (20S)-3β-(Dimethylamino)-4,4,14-trimethyl-20-(methylamino)-B(9a)-homo-19-nor-5α-pregna-9(11),9a-dien-16α-ol Chemical Properties |
| | (20S)-3β-(Dimethylamino)-4,4,14-trimethyl-20-(methylamino)-B(9a)-homo-19-nor-5α-pregna-9(11),9a-dien-16α-ol Usage And Synthesis |
| Description | This alkaloid occurs in Buxus sernpervirens var. bullatus and forms colourless
needles from Me2CO. It is dextrorotatory with a specific rotation of [α]D25 + 20°
(c 0.59, MeOH) and gives an ultraviolet spectrum in EtOH with absorption
maxima at 236, 246, 255 and 273 nm. Treatment with methyl iodide yields
buxaminol A. | | References | Bauerova, Paulik, Collect. Czech. Chern. Cornrnun., 40, 3055 (1975) |
| | (20S)-3β-(Dimethylamino)-4,4,14-trimethyl-20-(methylamino)-B(9a)-homo-19-nor-5α-pregna-9(11),9a-dien-16α-ol Preparation Products And Raw materials |
|