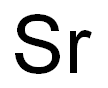How funny strontium flame test
Jan 24,2024
The flame test is an analytical chemistry method used to help identify metal ions.It is all about thermal energy, electrons, and the energy of photons.
Strontium flame test

Positive result of a flame test for strontium (Sr), producing a red colour.
This is due to the excitation of electrons in the strontium by the heat of the flame. As these electrons lose their energy, they emit photons of red light. The colour of the flame is different for different elements and can be used to identify unknown substances. The metal compound being tested here is strontium chloride.
- Related articles
- Related Qustion
- Strontium Industrial Applications and Uses Sep 25, 2019
Strontium chemical symbol Sr, atomic number 38, and relative atomic mass (i.e., atomic weight) 87.62, is the fourth alkaline-earth metal, i.e., elements of group IIA of Mendeleev’s periodic chart. The word strontium comes from Strontian, a
1,9-decadiene has a molecular formula C10H18, and sterling is a colorless liquid. This compound is widely used in organic synthesis as an important organic intermediate.....
Jan 24,2024Organic reagentsThe flame test is an analytical chemistry method used to help identify metal ions.The flame color of lithium chloride is a dark red flame.....
Jan 24,2024Inorganic chemistrySTRONTIUM
7440-24-6You may like
- What is nitrogen used for?
May 24, 2024
- The structure of Aluminum oxide
May 24, 2024
- The brief introduction of Lead selenide
May 23, 2024