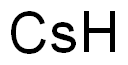Mechanism of Cesium
Dec 30,2021
Cesium was discovered in 1860 by Robert Bunsen and Gustav Kirchoff. It is used in the most accurate atomic clocks. Cesium melts at 28.41°C (just below body temperature) and occurs in Earth’s crust at 2.6 ppm. Cesium is the rarest of the naturally occurring alkali metals as the isotope 133Cs. Its compounds are correspondingly rare. Granites contain about 1 ppm cesium and sedimentary rocks contain approximately 4 ppm cesium. The most common commercial source of cesium is pollucite, which contains between 5 and 32% cesium oxide. Radioactive forms of cesium (134Cs and 137Cs) can also be found in the environment. They are produced during nuclear fission, and are used in cancer treatment.

Uses
Cesium is used in photovoltaic cells, vacuum tubes, scintillation counters, and atomic clocks.
Environmental Fate
Naturally occurring cesium can enter the environment mostly from the erosion and weathering of rocks and minerals. The production and use of cesium compounds may also result in their release to the environment through various waste streams. However, there are relatively few commercial uses for cesium compounds, such as cesium radioactive isotopes (134Cs and 137Cs), and they have been released into the environment by human activities such as the atmospheric testing of nuclear weapons (1945–80) and leakages at nuclear power plants. Cesium compounds can travel long distances in the air before being brought back to the earth by rainfall and gravitational settling. If released to water, cesium compounds are deposited on land and water via wet and dry deposition. These deposited-cesium particles may be resuspended into the atmosphere from soil and dust. If released to soil, cesium compounds have low mobility and do not migrate below 40 cm in depth.
The majority of cesium ions are retained in the upper 20 cm of the soil surface. Clay and zeolite minerals strongly bind cesium cations irreversibly. Soils rich in organic matter also adsorb cesium ions. However, cesium compounds are readily exchangeable and highly available for plant uptake in these soils. If released into water, cesium compounds are very water soluble and exist primarily as cesium cations. Because most cesium compounds are ionic, they will not volatilize from water surfaces. Most cesium compounds released to water adsorb to suspended solids in the water column and ultimately they are deposited in sediments. Cesium compounds bioconcentrate and have been shown to bioaccumulate in both terrestrial and aquatic food chains. The half-life of 134Cs is ~2 years and that of 137Cs is~30 years.
Mechanism
Stable cesium was shown to affect various central nervous system functions, mainly involving displacing potassium, with which it competes for transport through the potassium channel, and it can also activate sodium pump and subsequent transport into the cell across membranes. Thus, this resulted in potassium deficiency.
Radioactive isotopes of cesium, such as 134Cs and 137Cs, are
a greater health concern than stable cesium. These radioactive
isotopes of cesium are formed during nuclear fission. Both134Cs and 137Cs emit beta and gamma radiations. Beta radiation
travels short distances and can penetrate the skin and
superficial body tissues, whereas gamma radiation can travel
great distances and penetrate the entire body. Both beta and
gamma radiations may induce tissue damage and disruption of
cellular function.
- Related articles
- Related Qustion
- Cesium:Discovery,Minerals,Chemistry,Uses,Hazards May 31, 2024
Cesium is a relatively rare element, about 3 ppm in the Earth’s crust. It is the 45th most abundant element and the 36th among the metals. It is 3.3% as abundant as Rb, with which it is closely associated, chemically.
- The applications and Preparation of Cesium Sep 27, 2019
Cesium, or Caesium, chemical symbol Cs, atomic number 55, and relative atomic mass (i.e., atomic weight) 132.90545, is the fifth alkali metal of group IA of Mendeleev’s periodic chart. The word cesium comes from the Latin word caesius, mean
- Facts About Cesium Sep 26, 2019
Cesium is a rare, silver-white, shiny metal with brilliant blue spectral lines; the element's name comes from "caesius," a Latin word meaning "sky blue." It is the softest metal, with a consistency of wax at room temperature. It would melt
Calcium chloride is an inorganic compound, a salt with the chemical formula CaCl2. It is a white colored crystalline solid at room temperature, and it is highly soluble in water. It can be created by neutralising hydrochloric acid with calc....
Dec 29,2021Inorganic chemistryChloral hydrate was first synthesized in 1832 by Justus von Liebig and was the first synthetic central nervous system (CNS) depressant. It was used to treat delirium tremens, insomnia, and anxiety, although it is considered an unapproved dr....
Dec 30,2021API