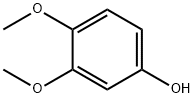
3,4-Dimethoxyphenol synthesis
- Product Name:3,4-Dimethoxyphenol
- CAS Number:2033-89-8
- Molecular formula:C8H10O3
- Molecular Weight:154.16
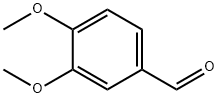
120-14-9
743 suppliers
$5.00/25g

2033-89-8
255 suppliers
$15.00/1g
Yield:2033-89-8 96%
Reaction Conditions:
Stage #1:3,4-dimethoxy-benzaldehyde with 3-chloro-benzenecarboperoxoic acid in dichloromethane at 20; for 15 h;
Stage #2: with potassium carbonate in methanol at 20; for 0.5 h;
Steps:
3,4-dimethoxyphenol (Compound 3)
Chloroperbenzoic acid (2.23 g with 77%, ca. 9.93 mmol) was added to a solution of 3,4-dimethoxybenzaldehyde (Compound 2) (1.50 g, 9.03 mmol) dissolved in CH2Cl2 (25 mL) Was stirred at room temperature for 15 hours. The reaction mixture was quenched by the addition of dimethyl sulfide (1 mL) and washed with a saturated aqueous solution of Na2SO3 (3 x 20 mL) and brine (20 mL). The organic solvent phase was dried over anhydrous Na2SO4 and concentrated in vacuo. The residue was dissolved in methanol (23 mL) and then treated with K2CO3 (2.49 g, 18.05 mmol) and the mixture was stirred at room temperature for 30 min. After removal of the methanol, the residue was dissolved in EtOAc (30 mL), washed with H2O (2 x 15 mL), brine (15 mL) and dried over anhydrous Na2SO4. The organic solvent phase was filtered and concentrated, and the residue was purified by column chromatography (EtOAc / hexane = 1/1) to give a white solid compound 3 (1.34 g, 96%).
References:
Hallym University Industry-Academic Cooperation Foundation;Jeon, Chung Gab;Kongara, Damodar KR101740915, 2017, B1 Location in patent:Paragraph 0053-0054
122775-35-3
322 suppliers
$5.00/1g

2033-89-8
255 suppliers
$15.00/1g

6315-89-5
304 suppliers
$7.00/10g

2033-89-8
255 suppliers
$15.00/1g

2033-88-7
0 suppliers
inquiry

2033-89-8
255 suppliers
$15.00/1g

67-56-1
731 suppliers
$7.29/5ml-f
62-44-2
500 suppliers
$19.00/25g

2033-89-8
255 suppliers
$15.00/1g

881-70-9
31 suppliers
$105.00/500mg
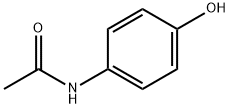
103-90-2
1043 suppliers
$9.00/1g