
2-AMINO-6-METHYL-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID ETHYL ESTER synthesis
- Product Name:2-AMINO-6-METHYL-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID ETHYL ESTER
- CAS Number:76981-71-0
- Molecular formula:C12H17NO2S
- Molecular Weight:239.33
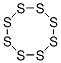
10544-50-0
21 suppliers
inquiry
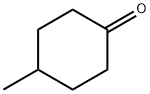
589-92-4
253 suppliers
$6.00/5g
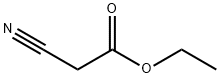
105-56-6
432 suppliers
$10.00/5ml
![2-AMINO-6-METHYL-4,5,6,7-TETRAHYDRO-BENZO[B]THIOPHENE-3-CARBOXYLIC ACID ETHYL ESTER](/CAS/GIF/76981-71-0.gif)
76981-71-0
83 suppliers
$32.20/1g
Yield:-
Reaction Conditions:
with 4-methyl-morpholine in neat (no solvent) at 75; for 1 h;Gewald Aminoheterocycles Synthesis;
Steps:
General Procedure for the Preparation of Alkyl 2-Aminobenzo[b]thiophene-3-carboxylates 7a-j, Exemplified with 7a:
General procedure: A mixture of cyclohexanone (0.196 g,2.0 mmol), methyl cyanoacetate (0.198 g, 2.0 mmol),elemental sulfur (0.064 g, 2.0 mmol) and morpholine (0.174g, 2.0 mmol) was heated at 75 °C for 1 h. After nearlycomplete conversion to the corresponding 2-aminothiophene, as was indicated by TLC monitoring, thereaction mixture was cooled to r.t. and the solid residue was recrystallized from EtOH to afford methyl 2-amino-4,5,6,7-tetrahydrobenzo[b]thiophene-3-carboxylate (6a). Then asolution of 6a (0.211 g, 1 mmol) in benzonitrile (2 mL) was heated within a flask equipped with an air-filled balloon at 200 °C for 24 h in a silicone oil bath. Progress of the reaction was monitored by TLC. After completion of the reaction, the mixture was cooled to r.t. and the excess of benzonitrile was removed under the reduced pressure. The crude product was purified by column chromatography using n-hexane-EtOAc(8:1) as eluent. The solvent was evaporated under the reduced pressure and the residue was crystallized from nhexane-EtOAc (5:1) to afford the pure product 7a as paleyellow crystals.
References:
Adib, Mehdi;Bayanati, Maryam;Soheilizad, Mehdi;Ghazvini, Helia Janatian;Tajbakhsh, Mahmood;Amanlou, Massoud [Synlett,2014,vol. 25,# 20,art. no. ST-2014-D0713-L,p. 2918 - 2922]