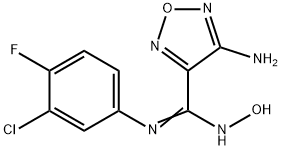
indoleaMine-2,3-dioxygenase inhibitor INCB024360 synthesis
- Product Name:indoleaMine-2,3-dioxygenase inhibitor INCB024360
- CAS Number:914471-09-3
- Molecular formula:C9H7ClFN5O2
- Molecular Weight:271.64
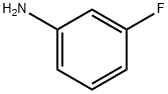
372-19-0
321 suppliers
$10.00/1g
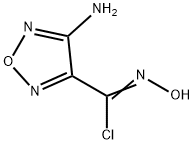
147085-13-0
65 suppliers
$7.00/250mg

914471-09-3
126 suppliers
$25.00/1mg
Yield: 91%
Reaction Conditions:
with sodium hydrogencarbonate in tetrahydrofuran at 45; for 3.16667 h;Reflux;
Steps:
4.1.4. Protocol for the synthesis of 4-amino-N-(3-bromo-4-fluorophenyl)-N'-hydroxy-1,2,5-oxadiazole-3-carboximidamide (8a)
General procedure: A 150 mL four-necked flask with a magnetic stir bar was chargedwith compound 7 (3.24 g, 20 mmol), sodium bicarbonate (3.36 g,40 mmol, 2 equiv) and tetrahydrofuran (70 mL). At 45 °C, a solutionof 3-bromo-4-fluoroaniline (4.18 g, 22 mol, 1.1 equiv) in tetrahydrofuran(15 mL)was added to the reaction mixture in 10 min. Thenthe reaction mixture was heat to reflux for another 3 h. Oncompleting of the reaction monitored by TLC, the sodium bicarbonatewas filtered and the filtrate was evaporated in vacuo. Theresidue was dissolved with ethyl acetate (70 mL), washed withwater (40 mL), brine, dried over anhydrous Na2SO4 and concentratedin vacuo. The crude product was slurried in petroleum ether(50 mL) for 1 h and the precipitate was collected by filtration anddried to give the desired product (5.75 g, 91%) as a white solid.
References:
Du, Qianming;Feng, Xi;Wang, Yinuo;Xu, Xi;Zhang, Yan;Qu, Xinliang;Li, Zhiyu;Bian, Jinlei [European Journal of Medicinal Chemistry,2019,vol. 182,art. no. 111629] Location in patent:supporting information
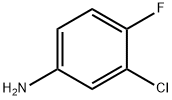
367-21-5
505 suppliers
$5.00/25g

147085-13-0
65 suppliers
$7.00/250mg

914471-09-3
126 suppliers
$25.00/1mg