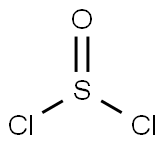
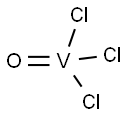
Vanadium(III) chloride
- CAS No.
- 7718-98-1
- Chemical Name:
- Vanadium(III) chloride
- Synonyms
- VCl3;VANADIUM CHLORIDE;VANADIUM TRICHLORIDE;trichlorovanadium;Vanadium(Ⅳ)chloride;Vanadium(Ⅲ)chloride;VANADIUM(III) CHLORIDE;vanadiumchloride(vcl3);Trichlorovanadium(III);Vanadium chloride (VCl3)
- CBNumber:
- CB0753498
- Molecular Formula:
- Cl3V
- Molecular Weight:
- 157.29
- MDL Number:
- MFCD00011454
- MOL File:
- 7718-98-1.mol
- MSDS File:
- SDS
| Melting point | 250 °C | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Density | 3 g/mL at 25 °C(lit.) | ||||||||||||||
| storage temp. | no restrictions. | ||||||||||||||
| solubility | Soluble in tetrahydrofuran, alcohol, ether and hydrocarbons. | ||||||||||||||
| form | Powder | ||||||||||||||
| Specific Gravity | 3. | ||||||||||||||
| color | Dark blue to violet to black | ||||||||||||||
| Water Solubility | decomposes in H2O; soluble alcohol, ether [HAW93] | ||||||||||||||
| Sensitive | Moisture Sensitive | ||||||||||||||
| Crystal Structure | BiI3 type | ||||||||||||||
| crystal system | Three sides | ||||||||||||||
| Space group | R3 | ||||||||||||||
| Lattice constant |
|
||||||||||||||
| Exposure limits | NIOSH: Ceiling 0.05 mg/m3 | ||||||||||||||
| CAS DataBase Reference | 7718-98-1(CAS DataBase Reference) | ||||||||||||||
| EWG's Food Scores | 1 | ||||||||||||||
| EPA Substance Registry System | Vanadium chloride (VCl3) (7718-98-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H302-H314 | |||||||||
| Precautionary statements | P260-P270-P280-P301+P312-P303+P361+P353-P305+P351+P338 | |||||||||
| Hazard Codes | C | |||||||||
| Risk Statements | 22-34 | |||||||||
| Safety Statements | 26-27-28-36/39-45 | |||||||||
| RIDADR | UN 2475 8/PG 3 | |||||||||
| WGK Germany | 3 | |||||||||
| RTECS | YW2800000 | |||||||||
| F | 3-10 | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | III | |||||||||
| HS Code | 28273990 | |||||||||
| Toxicity | LD50 orally in Rabbit: 350 mg/kg | |||||||||
| NFPA 704 |
|
Vanadium(III) chloride price More Price(24)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | 208272 | Vanadium(III) chloride 97% | 7718-98-1 | 1g | $39.2 | 2024-03-01 | Buy |
| Sigma-Aldrich | 208272 | Vanadium(III) chloride 97% | 7718-98-1 | 5g | $48.8 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1.12393 | Vanadium(III) chloride 99+ | 7718-98-1 | 25g | $180 | 2024-03-01 | Buy |
| Alfa Aesar | 014039 | Vanadium(III) chloride 99% (metals basis) | 7718-98-1 | 10g | $103 | 2024-03-01 | Buy |
| Alfa Aesar | 014039 | Vanadium(III) chloride 99% (metals basis) | 7718-98-1 | 50g | $443 | 2023-06-20 | Buy |
Vanadium(III) chloride Chemical Properties,Uses,Production
Chemical Properties
Vanadium(III) chloride is pink, deliquescent crystals that decompose upon heating. It is soluble in absolute alcohol and ether but decomposes in water. The hydrated ions of trivalent vanadium are green. They form hydrated coordinated chlorides with excess chloride ions and react with liquid ammonia to produce ammine compounds. They also react with gaseous ammonia to form nitrides, with amines and other organic compounds to produce corresponding coordination compounds, and with certain aromatic hydroxy acids to exhibit characteristic color reactions.
Uses
Preparation of vanadium dichloride and organovanadium compounds.
Uses
Vanadium(III) chloride has been used as a catalyst for Biginelli condensation. As a reducing agent, it is capable of converting nitrate to nitrite in water samples, allowing for the direct detection of nitrates.
Preparation
Vanadium trioxide powder (2.1 g) and 8.5 ml of pure SOCl2 are placed in a bomb tube about 1.5 cm. in diameter, and the sealed tube is heated for 24 hours at 200℃. The tube is cooled to below 0℃ (in order to lower the SO2 vapor pressure) and is then opened to allow SO2 to escape. Then the tube contents are flushed out, under anhydrous conditions, into a small flask, using some SOCl2 for this purpose (the SOCl2 is then removed by vacuum distillation). The VCl3 residue is washed several times with very pure CS2 to remove traces of S2Cl2, and then thoroughly vacuum-dried at 80℃. Fine crystals of dark-violet VCl3 are obtained in nearly quantitative yield.
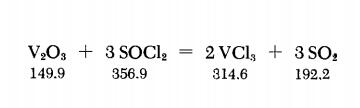
General Description
Vanadium(III) chloride is a crystalline solid with variable oxidation states. It is widely used as a catalyst in organic synthesis, polymerization processes, and redox batteries. It acts as a reducing agent.
Air & Water Reactions
Deliquescent. Generates acid mists when exposed to moist air. Dissolves in water with formation of an acidic solution and generation of acidic fumes.
Reactivity Profile
VANADIUM(III) CHLORIDE attacks many organic compounds. Can catalyze organic reactions. Aqueous solutions react as acids to neutralize bases. These neutralizations generate heat, but less than is generated by neutralization of inorganic acids, inorganic oxoacids, and carboxylic acid. Combination of the trichloride with methylmagnesium iodide, or other Grignard type reagents, can be violently explosive under a variety of conditions, Chem. Rev., 1955, 55, 560.
Health Hazard
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Reaction with water or moist air will release toxic, corrosive or flammable gases. Reaction with water may generate much heat that will increase the concentration of fumes in the air. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
Fire Hazard
Non-combustible, substance itself does not burn but may decompose upon heating to produce corrosive and/or toxic fumes. Vapors may accumulate in confined areas (basement, tanks, hopper/tank cars etc.). Substance will react with water (some violently), releasing corrosive and/or toxic gases and runoff. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated or if contaminated with water.
Flammability and Explosibility
Not classified
Safety Profile
Poison by ingestion and subcutaneous routes. A corrosive irritant to skin, eyes, and mucous membranes. Extremely violent reaction with methyl magnesium iodide and other Grignard reagents. When heated to decomposition it emits toxic fumes of VOx and Cl-. See also VANADIUM COMPOUNDS and HYDROCHLORIC ACID.
References
[1] BERNHARD SCHNETGER Carola L. Determination of nitrate plus nitrite in small volume marine water samples using vanadium(III)chloride as a reduction agent[J]. Marine Chemistry, 2014, 160: Pages 91-98. DOI:10.1016/j.marchem.2014.01.010.
[2] XU Toshikazu H. Vanadium-Catalyzed Pinacol Coupling Reaction in Water[J]. The Journal of Organic Chemistry, 2005, 70 21: 8594-8596. DOI:10.1021/jo051213f.
[3] GOWRAVARAM SABITHA. Vanadium(III) chloride: A mild and efficient catalyst for the chemoselective deprotection of acetonides[J]. Journal of Molecular Catalysis A: Chemical, 2005, 238 1: Pages 229-232. DOI:10.1016/j.molcata.2005.05.028.
[4] SHU WANG. Automated determination of nitrate plus nitrite in aqueous samples with flow injection analysis using vanadium (III) chloride as reductant.[J]. Talanta, 2016, 47 1: 744-748. DOI:10.1016/j.talanta.2015.06.031.
Vanadium(III) chloride Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21639 | 55 |
| Hubei xin bonus chemical co. LTD | 86-13657291602 | linda@hubeijusheng.com | CHINA | 22963 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 | sales@chemdad.com | China | 39894 | 58 |
| Antai Fine Chemical Technology Co.,Limited | 18503026267 | info@antaichem.com | CHINA | 9636 | 58 |
| Career Henan Chemica Co | +86-0371-86658258 +8613203830695 | laboratory@coreychem.com | China | 30241 | 58 |
| Hefei TNJ Chemical Industry Co.,Ltd. | +86-0551-65418671 +8618949823763 | sales@tnjchem.com | China | 34553 | 58 |
| Shaanxi Didu New Materials Co. Ltd | +86-89586680 +86-13289823923 | 1026@dideu.com | China | 8670 | 58 |
| LEAP CHEM CO., LTD. | +86-852-30606658 | market18@leapchem.com | China | 24727 | 58 |
| Shanghai Acmec Biochemical Technology Co., Ltd. | +undefined18621343501 | product@acmec-e.com | China | 33338 | 58 |
| Aladdin Scientific | +1-+1(833)-552-7181 | sales@aladdinsci.com | United States | 52925 | 58 |
View Lastest Price from Vanadium(III) chloride manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2021-08-12 | VANADIUM(III) CHLORIDE
7718-98-1
|
US $15.00-10.00 / KG | 1KG | 99%+ HPLC | Monthly supply of 1 ton | Zhuozhou Wenxi import and Export Co., Ltd |
-

- VANADIUM(III) CHLORIDE
7718-98-1
- US $15.00-10.00 / KG
- 99%+ HPLC
- Zhuozhou Wenxi import and Export Co., Ltd