Aliskiren hemifumarate
- CAS No.
- 173334-58-2
- Chemical Name:
- Aliskiren hemifumarate
- Synonyms
- HSDB 7272;CCRIS 9267;Unii-C8A0p8G029;ALISKIREN FUMARATE;skiren heMifuMarate;Rasilez HeMifuMarate;SPP 100 hemifumarate;Tekturna HeMifuMarate;Aliskiren hemifumarate;Aliskiren hemifumerate
- CBNumber:
- CB21471023
- Molecular Formula:
- C30H53N3O6
- Molecular Weight:
- 551.75832
- MDL Number:
- MFCD10566724
- MOL File:
- 173334-58-2.mol
- MSDS File:
- SDS
| Melting point | 72-75?C |
|---|---|
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | Methanol (Slightly), Water (Slightly) |
| form | Solid |
| color | White |
| FDA UNII | C8A0P8G029 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS08,GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H360-H335-H315-H319 | |||||||||
| Precautionary statements | P264-P280-P302+P352-P321-P332+P313-P362-P264-P280-P305+P351+P338-P337+P313P | |||||||||
| NFPA 704 |
|
Aliskiren hemifumarate price More Price(49)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | SML2077 | Aliskiren hemifumarate ≥95% (HPLC) | 173334-58-2 | 10MG | $64.9 | 2024-03-01 | Buy |
| Sigma-Aldrich | SML2077 | Aliskiren hemifumarate ≥95% (HPLC) | 173334-58-2 | 50MG | $211 | 2024-03-01 | Buy |
| Cayman Chemical | 19640 | Aliskiren (hemifumarate) ≥95% | 173334-58-2 | 1mg | $32 | 2024-03-01 | Buy |
| Cayman Chemical | 19640 | Aliskiren (hemifumarate) ≥95% | 173334-58-2 | 5mg | $116 | 2024-03-01 | Buy |
| Cayman Chemical | 19640 | Aliskiren (hemifumarate) ≥95% | 173334-58-2 | 10mg | $214 | 2024-03-01 | Buy |
Aliskiren hemifumarate Chemical Properties,Uses,Production
Description
Aliskiren hemifumarate (Tekturna(R)) was the first renin inhibitor approved by FDA in March 2007, and genomic analysis validated that Pgx-based dosing guidelines are not required for this drug. The once-daily, oral, direct renin inhibitor received FDA approval for treatment of high blood pressure as mono therapy or in combination with other antihypertensive medications. Furthermore, aliskiren demonstrated increased efficacy when used in combination with other commonly used blood pressure-lowering medications. Novartis is conducting a large outcome trial program to evaluate the long-term effects of aliskiren and of direct renin inhibition in general.
Chemical Properties
white to slightly yellowish crystalline powder. It is soluble in phosphate buffer, n-Octanol, and highly soluble in water.
Uses
An orally active, synthetic nonpeptide renin inhibitor. Antihypertensive.
Uses
Aliskiren hemifumarate is a potent renin inhibitor (IC50 = 0.6 and 80 nM for human and rat respectively). It has antihypertensive activity, decreasing plasma renin activity and inhibiting the conversion of angiotensinogen to Angiotensin I by binding to the S3 sub-pocket of renin. Exhibits selectivity for renin over a range of other aspartic proteinases (>5000 nM). Lowers blood pressure in a hypertensive rodent model. Orally active.
Aliskiren hemifumarate (ALS) has been used to treat hypertension, alone or with other antihypertensive medications. It is suitable for oral administration. ALS regulates baseline systolic and diastolic blood pressure by blocking the catalytic activity of renin system at its rate-limiting step.
Definition
ChEBI: Aliskiren fumarate is the hemifumarate salt of aliskiren. It has a role as an antihypertensive agent. It contains an aliskiren.
Clinical Use
Renin inhibitor:
Hypertension
Synthesis
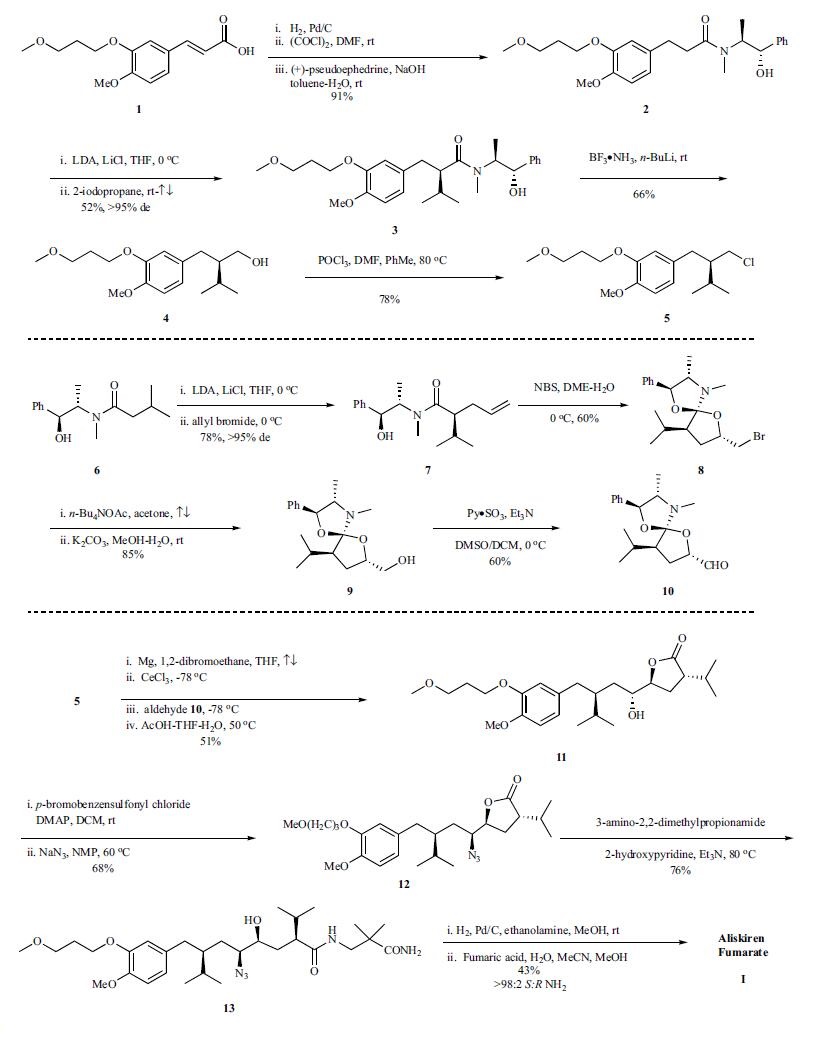
The synthesis of aliskiren by Novartis is depicted in the scheme.Aliskiren (I) was synthesized through a convergent synthetic strategy by coupling key intermediate chloride 5 with aldehyde 10. Hydrogenation of cinnamic acid 1, followed by generation of the acid chloride of the corresponding acid and reaction with (+)-pseudoephedrine provided amide 2 in 91% yield. Deprotonation of amide 2 with LDA followed by alkylation with 2-iodopropane in refluxing THF gave 3 as a single diastereomer in 52% yield. Reduction of the amide functionality in 3 using n-butyl lithium boron trifluoride ammonium complex proceeded without epimerization of the chiral center to give alcohol 4 in 66% yield. Chlorination of 4 using phosphorus oxychloride gave chloride 5, in 78% yield as the organometallic precursor for the eventual coupling to aldehyde 10. Synthesis of fragment 10 commenced with (+)-pseudoephedrine isovaleramide 6, which was efficiently deprotonated with LDA and alkylated using allyl bromide; diastereomerically pure 7 was obtained upon crystallization of the crude reaction mixture in 78% yield. Bromolactonization of 7, using n-bromosuccinimide in the absence of acetic acid gave amide acetal 8 with a single configuration at the spirocenter and a 6:1 mixture of trans:cis ring substituents. Displacement of the bromide using tetrabutylammonium acetate followed by basic hydrolysis provided alcohol 9 in 85% yield. Oxidation of 9 using dimethyl sulfoxide-sulfur trioxide/pyridine proceeded without epimerization to furnish the masked lactone aldehyde 10 in 60% yield. Coupling of fragments 5 and 10 was achieved by treatment of 10 with the organocerium reagent of the corresponding Grignard reagent prepared from 5. Hydrolysis of the crude spirocyclic addition product revealed that the hydroxylactone 11 was formed in 51% overall yield as an inseparable epimeric mixture with a Felkin-Anh selectivity of 85:15. The requisite nitrogen functionality was installed via the brosylate to give azido lactone 12 in 68% yield. Aminolysis with 3-amino-2,2-dimethylpropionamide led to formation of the open chain azido alcohol 13 in 76% yield. The synthesis of aliskiren was completed by azide hydrogenolysis and formation of the hemifumarate salt. Generation of pure aliskiren was achieved via crystallization which removed the residual minor (R)-epimer carried through from the Grignard addition step to afford aliskiren (I) in 43% yield.
Drug interactions
Potentially hazardous interactions with other drugs
Other antihypertensive agents: enhanced
antihypertensive effect; concentration possibly
reduced by irbesartan; increased risk of
hyperkalaemia and hypotension with ACE-Is and
ARBs.
Antifungals: concentration increased by itraconazole
and ketoconazole, avoid with itraconazole.
Ciclosporin: concentration of aliskiren increased -
avoid.
Diuretics: may reduce concentration of furosemide;
hyperkalaemia with potassium-sparing diuretics.
Grapefruit juice: concentration of aliskiren reduced
- avoid.
Heparins: increased risk of hyperkalaemia.
Potassium salts: increased risk of hyperkalaemia.
Metabolism
Approximately 1.4% of the total oral dose is metabolised by CYP3A4. Approximately 0.6% of the dose is recovered in urine following oral administration. Aliskiren is mainly eliminated as unchanged compound in the faeces (78%).
storage
Store at -20°C
Aliskiren hemifumarate Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Hebei Weibang Biotechnology Co., Ltd | +8615531157085 | abby@weibangbio.com | China | 8811 | 58 |
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 | sales@hbmojin.com | China | 12840 | 58 |
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 | deasea125996@gmail.com | China | 2472 | 58 |
| Nanjing Gold Pharmaceutical Technology Co. Ltd. | 025-84209270 15906146951 | CHINA | 115 | 55 | |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21634 | 55 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29882 | 58 |
| Biochempartner | 0086-13720134139 | candy@biochempartner.com | CHINA | 965 | 58 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28172 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-86-5926051114 +8618959220845 | sales@amoychem.com | China | 6383 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19553 | 58 |
View Lastest Price from Aliskiren hemifumarate manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-11-22 | Aliskiren hemifumarate
173334-58-2
|
US $0.00 / g | 1g | 98%min | 1000g | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2024-11-19 | Aliskiren hemifumarate
173334-58-2
|
US $50.00-98.00 / mg | 99.7% | 10g | TargetMol Chemicals Inc. | ||
 |
2024-11-12 | Aliskiren hemifumarate
173334-58-2
|
US $10.00 / KG | 1KG | 99% | 10 mt | Hebei Weibang Biotechnology Co., Ltd |
-

- Aliskiren hemifumarate
173334-58-2
- US $0.00 / g
- 98%min
- WUHAN FORTUNA CHEMICAL CO., LTD
-

- Aliskiren hemifumarate
173334-58-2
- US $50.00-98.00 / mg
- 99.7%
- TargetMol Chemicals Inc.
-

- Aliskiren hemifumarate
173334-58-2
- US $10.00 / KG
- 99%
- Hebei Weibang Biotechnology Co., Ltd





