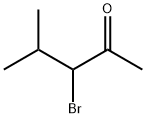
Preparation
Synthesis of 3-bromo-4-methyl-pentan-2-one: Br2 (16.14 g, 101.0 mmol) dissolved in acetic acid (10 mL) was added dropwise to a solution of isobuthylmethylketone (10.01 g, 100.0 mmol) dissolved in acetic acid (200 mL) at r.t. The solution was stirred overnight at r.t. Water (400 mL) was added and the solution was extracted with DCM (4 x 80 mL). The organic phase was washed with NaHCO3 (aq) (2 x 50 mL) and water (2 x 50 mL). The solvent was removed under reduced pressure giving the crude product (18.0 g) brown oil. Distillation gave 4.3 g (24 %) of the bromoketone 23(b.p. 60 – 62°C, 12mmHg).

Rf 0.48 in 15% EtOAc in Hexanes.
IR (neat) ν 2967, 2874, 1721, 1467, 1389, 1370, 1289, 1190, 1148, 1041, 948, 827 cm-1 . 1H NMR (300 MHz, CDCl3)a 0.96 (d, J = 6.3 Hz, 3 H), 1.11 (d, J = 6.6 Hz, 3 H), 2.14 - 2.30 (m, 1 H), 2.35 (s, 3 H), 4.03 (d, J = 8 Hz, 2 H). C 13 NMR (75 MHz, CDCl3) a 20.1, 26.3, 31.1, 63.0, 202.1.