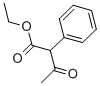
Ethyl 2-phenylacetoacetate
- CAS No.
- 5413-05-8
- Chemical Name:
- Ethyl 2-phenylacetoacetate
- Synonyms
- ETHYL 3-OXO-2-PHENYLBUTANOATE;2-Phenylacetoacetic acid ethyl ester;2-Phenylacetoacetate;ethyl 2-phenyl-3-oxobutanoate;Benzeneacetic acid, a-acetyl-, ethyl ester;a-acetyl-;99% Pure BMK;Bmk powder/oil;Ethyl 2-phenylacetoa;BMK Glycidate BMK Powder
- CBNumber:
- CB4772521
- Molecular Formula:
- C12H14O3
- Molecular Weight:
- 206.24
- MDL Number:
- MFCD00040490
- MOL File:
- 5413-05-8.mol
- MSDS File:
- SDS
| Melting point | 140-144°C/10mm |
|---|---|
| Boiling point | 140-144°C 10mm |
| Density | 1,085 g/cm3 |
| refractive index | 1.5130 |
| Flash point | 140-144°C/10mm |
| solubility | Chloroform (Slightly), Methanol (Slightly) |
| form | Oil |
| pka | 10.69±0.46(Predicted) |
| color | Colourless to Pale Brown |
| BRN | 1912655 |
| InChI | InChI=1S/C12H14O3/c1-3-15-12(14)11(9(2)13)10-7-5-4-6-8-10/h4-8,11H,3H2,1-2H3 |
| InChIKey | PWRUKIPYVGHRFL-UHFFFAOYSA-N |
| SMILES | C(C(=O)C)(C(=O)OCC)C1C=CC=CC=1 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H315-H319-H335 | |||||||||
| Precautionary statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 | |||||||||
| Safety Statements | 24/25 | |||||||||
| NFPA 704 |
|
Ethyl 2-phenylacetoacetate Chemical Properties,Uses,Production
Uses
Ethyl 2-Phenylacetoacetate is used in preparation of iridium polysubstituted quinoline diketonate complex and application as OLED.
Synthesis
A flame-dried flask was charged with 10 mmol (1 equiv) of sublimed potassium tert-butoxide in anhydrous DMF (50 mL) at room temperature under argon. Then, 10 mmol (1 equiv) of freshly distilled EAA was added to the reaction mixture and stirred for 30 min at 0 °C, followed by dropwise addition of diaryliodonium salt (4 mmol, 0.4 equiv to EAA) in 10 mL of DMF. Reaction was left stirring at room temperature for the time mentioned in the table. After confirming complete consumption of iodonium salt (by LCMS), to the reaction mixture was added 1 M HCl in one portion to bring the pH to around 5.0. The crude was extracted with diethyl ether until the aqueous layer was devoid of product. The organic layer was dried over sodium sulfate, and solvent was removed in vacuo. The product was purified by flash column chromatography (0.5-2% of hexane in ethyl acetate). Ethyl 2-phenylacetoacetate 1H NMR (500 MHz, CDCl3) δ 13.13 (s, 0.3H), 7.41-7.27 (m, 4H), 7.18-7.13 (m, 1H), 4.69 (s, 0.7H), 4.27-4.15 (m, 2H), 2.19 (s, 2H), 1.86 (s, 1H), 1.28 (t, J = 7.1 Hz, 2H), 1.18 (t, J = 7.1 Hz, 1H). 13C NMR (126 MHz, CDCl3) δ 201.7, 174.0, 172.7, 168.6, 135.4, 132.8, 131.4, 129.4, 129.0, 128.4, 128.1, 127.0, 104.5, 65.9, 61.8, 60.8, 28.9, 20.0, 14.3, 14.2. HRMS (ESI-TOF) m/z: [M + H]+ calcd for C12H14O3 207.1016; found 207.1018.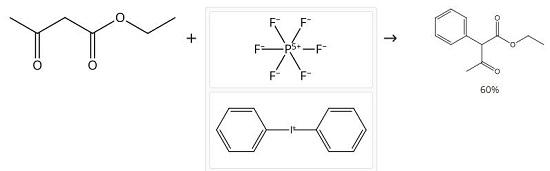
Ethyl 2-phenylacetoacetate Preparation Products And Raw materials
Raw materials
Preparation Products
5413-05-8(Ethyl 2-phenylacetoacetate)Related Search:
1of4