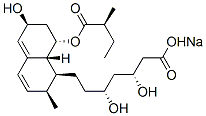
Pravastatin sodium
- CAS No.
- 81131-70-6
- Chemical Name:
- Pravastatin sodium
- Synonyms
- Prava;cs-514;Vasten;ELISOR;Lipidal;sq31000;Liprevil;Pravacol;Pravasin;Selipran
- CBNumber:
- CB9317123
- Molecular Formula:
- C23H36NaO7
- Molecular Weight:
- 447.52
- MDL Number:
- MFCD00887601
- MOL File:
- 81131-70-6.mol
- MSDS File:
- SDS
| Melting point | 171.2-173 °C |
|---|---|
| storage temp. | 2-8°C |
| solubility | H2O: 19 mg/mL |
| form | powder |
| color | white |
| Merck | 14,7714 |
| BCS Class | 1 (CLogP), 3 (LogP) |
| Stability | Hygroscopic |
| CAS DataBase Reference | 81131-70-6(CAS DataBase Reference) |
| NCI Dictionary of Cancer Terms | Pravachol; pravastatin sodium |
| FDA UNII | 3M8608UQ61 |
| NCI Drug Dictionary | Pravachol |
| Proposition 65 List | Pravastatin sodium |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|---|---|
| Signal word | Warning |
| Hazard statements | H315-H319-H335 |
| Precautionary statements | P261-P264-P271-P280-P302+P352-P304+P340-P305+P351+P338-P312-P321-P332+P313-P337+P313-P362-P403+P233-P405-P501 |
| Hazard Codes | F,C |
| Risk Statements | 11-34 |
| Safety Statements | 16-26-36/37/39-45 |
| RIDADR | 3077 |
| WGK Germany | 2 |
| RTECS | QJ7185000 |
| HazardClass | 9 |
| PackingGroup | III |
| HS Code | 29329990 |
Pravastatin sodium price More Price(50)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | P4498 | Pravastatin sodium salt hydrate ≥98% (HPLC), powder | 81131-70-6 | 25mg | $92.7 | 2024-03-01 | Buy |
| Sigma-Aldrich | 524403 | Pravastatin, Sodium Salt - CAS 81131-70-6 - Calbiochem A water-soluble, competitive 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor that potently blocks in vivo cholesterol synthesis (Ki = ~1 nM). | 81131-70-6 | 25mg | $130 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1554206 | Pravastatin sodium | 81131-70-6 | 50mg | $474 | 2024-03-01 | Buy |
| TCI Chemical | P1796 | Pravastatin Sodium >98.0%(HPLC)(T) | 81131-70-6 | 100mg | $314 | 2024-03-01 | Buy |
| TCI Chemical | P1796 | Pravastatin Sodium >98.0%(HPLC)(T) | 81131-70-6 | 500mg | $1059 | 2024-03-01 | Buy |
Pravastatin sodium Chemical Properties,Uses,Production
Chemical Properties
White Crystalline Powder
Originator
Apo-Pravastatin ,Apotex Inc., Canada
Uses
An HMGCR inhibitor which blocks cholesterol synthesis
Uses
antihyperlipidemic, HMGCoA reductase inhibitor
Uses
Pravastatin sodium is the salt of pravastatin, a ring-opened member of the statin family. Pravastatin is produced biosynthetically from compactin (mevastatin) by a number of microorganisms, notably Absidia, Cunninghamella, Syncephalastrum, Nocardia or Streptomyces. Typically, statins like compactin, lovastatin and simvastatin possess a β-hydroxy lactone ring which is a pro-drug for the readily ring-opened dihydroxyacid generally regarded as the active HMG-CoAreductase inhibitor. Importantly, the free carboxylic acid enables pravastatin to be freely water soluble within biological pH ranges.
Uses
A competitive inhibitor of HMG-CoA reductase. Bioactive metabolite of Mevastatin
Definition
ChEBI: An organic sodium salt that is the sodium salt of pravastatin. A reversible inhibitor of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA), it is used for lowering cholesterol and preventing cardiovascular disease. It is one of the lower potency statins, but has the advantage of fewer side effects compared with lovastatin and simvastatin.
Manufacturing Process
Pravastatin was isolated as products of enzymatic hydroxylation by some kinds of microorganisms of [1S-[1-α(R*),7β,8β(2S*,4S*)8αβ]]-2methylbutanoic acid 1,2,3,7,8,8a-hexahydro-7-methyl-8-[2-(tetrahydro-4hydroxy-6-oxo-2))-pyran-2-yl)ethyl]-1-naphthalenic lactone (campactin) or their carboxylic acid or their salts (products of animal metabolism of microorganisms from the genera Nocardia, Streptomyces et cetera).
Pravastatin may be preparated by using the microorganisms of genera Nocardia (method 1) and Mortierella (method 2).
Method 1
Cultivation of Nocardia autotrophica subsp. amethystineCells of Nocardia autotrophica subsp. amethystina FERM P-6183 was inoculated from a slant culture by means of a platinum loop into each of twenty 500 ml Erlenmeyer flasks, each containing 100 ml of a culture medium having the following composition: glucose - 1.0%, peptone - 0.2%, meat extract - 0.1%, yeast extract - 0.1%, corn steep liquor 0.3%, tap water balance..
Shaking was then carried out at 26°C and 220 r.p.m. for 2 days, at which time sodium 2-methyl-8-(2-methyl-1-oxobutoxy)-β,δ-dihydroxy(1S-(1-α(βS*,δ-S*),2-α,6-α,8-β(R*),8a-α))-1-1,2,6,7,8,8ahexahydronaphthaleneheptanoate was added to a final concentration of 0.05% w/v. Incubation was continued at 26°C and 220 r.p.m. for a further 5 days.
Preparation of pravastatin
After completion of the cultivation, the reaction mixture was filtered and the pH of the filtrate was adjusted to a value of 3 by the addition of trifluoroacetic acid. The acidified filtrate was then extracted three times, each with 1 liter of ethyl acetate, to give extracts containing a mixture (6-α and 6-β) of (1S-(1α,β- S*,δ-S*),2-α,8-βR*),8a-α))-1-naphthaleneheptanoic acid 1,2,6,7,8,8ahexahydro-2-methyl-8-(2-methyl-1-oxobutoxy)-β,δ,6-trihydroxy.
This extract was then immediately transferred into a 5% w/v aqueous solution of sodium hydrogen carbonate, and the pH of the mixture was adjusted to a value of 7.0 by the addition of 2 N hydrochloric acid. The mixture was then adsorbed on a Diaion HP-20 column. The column was washed with water and then eluted with 50% v/v aqueous acetone to give a fraction containing (1S(1-α,β- S*,δ-S*),2-α,6-α,8-βR*),8a-α))-1-naphthaleneheptanoic acid 1,2,6,7,8,8a-hexahydro-2-methyl-8-(2-methyl-1-oxobutoxy)-β,δ,6-trihydroxy-, monosodium salt (pravastatin). This was freeze-dried, to give 200 mg of pravastatine.
Method 2
Cultivation of Mortierella maculata nov. spec. E-97 [NCAIM(P)F 001266]
A spore suspension was prepared with 5 ml of a 0.9% sodium chloride solution obtained from a 7-10 day old, malt extract-yeast extract agar slant culture of Mortierella maculata nov. spec. E-97 [NCAIM(P)F 001266] strain able to 6-β-hydroxylate compactin and the suspension was used to inoculate 100 ml inoculum medium PI (glucose-50 g, soybean meal-20 g, in 1000 ml tap water) sterilized in a 500 ml Erlenmeyer flask.
5 liters working volume a bioconversion culture medium is prepared (glucose20 g, glycerine-20 g, soybean meal-20 g, peptone-5 g, potassium dihydrogen phosphate-0.5 g, polypropylene glycol 2000-1 g, in 1000 ml tap water); the components of the culture medium are added corresponding to 5 liters. Then it was sterilized for 45 min at 121°C and seeded with 500 ml of the inoculum culture.
Before sterilization the pH of the medium was adjusted to 7.0 value.
The fermentation was carried out at 28°C, with a stirring rate of 400 rpm andwith an aeration rate from bottom direction 60 liters/hour for 4 days. At the 2nd day after the transfer the culture started to foam heavily, which can be decreased by the addition of further polypropylene glycol 2000. The pH reached 6.3-7.5 by the 4th day. The feeding of the sodium 2-methyl-8-(2methyl-1-oxobutoxy)-β,δ-dihydroxy(1S-(1-α(β- S*,δ-S*),2-α,6-α,8-β(R*),8aα))-1-1,2,6,7,8,8a-hexahydronaphthaleneheptanoate substrate is allowed to be started if the pH of the broth is above 6.3.
Preparation of pravastatinAt the 4th day of the fermentation 2.5 g compactin substrate is added in sterile filtered aqueous solution. Calculated for the volume of the broth 0.51.0% glucose was added into the culture depending on the pH in the form of 50% solution sterilized at 121°C for 25 min in parallel with the substrate feeding. After 24 hours the compactin substrate is consumed from the culture (is detected by HPLC) and was converted to pravastatin. By lyophilization of the aqueous residue 1.3 g pravastatin was obtained. The chromatographically pure product was crystallized from a mixture of ethanol and ethyl acetate. Melting point: 170-173°C (decomp.).
brand name
Pravachol (Bristol-Myers Squibb).
Therapeutic Function
Antihyperlipidemic
General Description
A water-soluble, competitive 3-hydroxy-3-methylglutaryl coenzyme A (HMG-CoA) reductase inhibitor that potently blocks in vivo cholesterol synthesis (Ki = ~1 nM). Offers cardioprotection. Also acts as an immunomodulator and an inhibitor of ras p21 isoprenylation.
Biological Activity
Water-soluble, competitive inhibitor of 3-hydroxy-3-methyl coenzyme A (HMG-CoA) reductase. Potently blocks cholesterol synthesis in vivo (K i ~ 1 nM) and displays cardioprotective properties.
Biochem/physiol Actions
Primary TargetAMG-CoA reductase
Clinical Use
HMG CoA reductase inhibitor:
Hypercholesterolaemia
Drug interactions
Potentially hazardous interactions with other drugs
Antibacterials: increased risk of myopathy with
daptomycin, fusidic acid (avoid) and telithromycin;
concentration increased by clarithromycin and
erythromycin.
Antivirals: increased risk of myopathy with
atazanavir and boceprevir; concentration possibly
increased by darunavir; concentration reduced by
efavirenz.
Ciclosporin: increased risk of myopathy.
Colchicine: possible increased risk of myopathy.
Lipid lowering agents: increased risk of myopathy
with fibrates, gemfibrozil (avoid) and nicotinic acid.
Metabolism
Pravastatin undergoes extensive hepatic metabolism to a relatively inactive metabolite. About 70% of an oral dose of pravastatin is excreted in the faeces, as unabsorbed drug and via the bile.
storage
Desiccate at RT
References
[1] bolego c, poli a, cignarella a, catapano al, paoletti r. novel statins: pharmacological and clinical results. cardiovasc drugs ther. 2002 may;16(3):251-7.
[2] keidar s, aviram m, maor i, oiknine j, brook jg. pravastatin inhibits cellular cholesterol synthesis and increases low density lipoprotein receptor activity in macrophages: in vitro and in vivo studies. br j clin pharmacol. 1994 dec;38(6):513-9.
[3] menter dg, ramsauer vp, harirforoosh s, chakraborty k, yang p, hsi l, newman ra, krishnan k. differential effects of pravastatin and simvastatin on the growth of tumor cells from different organ sites. plos one. 2011;6(12):e28813.
Pravastatin sodium Preparation Products And Raw materials
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 | sales01@cooperate-pharm.com | CHINA | 1803 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21634 | 55 |
| Hubei Jusheng Technology Co.,Ltd. | 18871490254 | linda@hubeijusheng.com | CHINA | 28172 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-86-5926051114 +8618959220845 | sales@amoychem.com | China | 6383 | 58 |
| HubeiwidelychemicaltechnologyCo.,Ltd | 18627774460 | faith@widelychemical.com | CHINA | 742 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19553 | 58 |
| Shanghai Standard Technology Co., Ltd. | 18502101150 | ft-sales@nature-standard.com | CHINA | 1923 | 58 |
| Shenzhen Excellent Biotech Co., Ltd. | 13480692018 | ramyan@ex-biotech.com | CHINA | 954 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 | sales@conier.com | China | 49374 | 58 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | factory@coreychem.com | China | 29811 | 58 |
View Lastest Price from Pravastatin sodium manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2024-11-19 | Pravastatin sodium
81131-70-6
|
US $40.00-66.00 / mg | 99.08% | 10g | TargetMol Chemicals Inc. | ||
 |
2024-11-19 | Pravastatin sodium
81131-70-6
|
US $40.00-66.00 / mg | 99.08% | 10g | TargetMol Chemicals Inc. | ||
 |
2023-02-24 | Pravastatin sodium
81131-70-6
|
US $0.00 / mg | 20mg | ≥98%(HPLC) | 10 g | Shanghai Standard Technology Co., Ltd. |
-

- Pravastatin sodium
81131-70-6
- US $40.00-66.00 / mg
- 99.08%
- TargetMol Chemicals Inc.
-

- Pravastatin sodium
81131-70-6
- US $40.00-66.00 / mg
- 99.08%
- TargetMol Chemicals Inc.
-
- Pravastatin sodium
81131-70-6
- US $0.00 / mg
- ≥98%(HPLC)
- Shanghai Standard Technology Co., Ltd.
81131-70-6(Pravastatin sodium)Related Search:
1of4