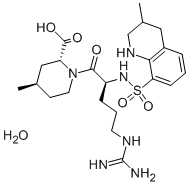
Argatroban
- CAS No.
- 141396-28-3
- Chemical Name:
- Argatroban
- Synonyms
- Argatroban Monohydrate;(2r,4r)-4-methyl-1-[n-[(3-methyl-1,2,3,4-tetrahydro-8-quinolinyl)sulfonyl]-l-arginyl]-2-piperidinecarboxylic acid;GN1600;AG, SAJ;CS-1966;US-DMF:035491;Argatroban API;Acova monohydrate;Argatroban (350 mg);API Cas:141396-28-3
- CBNumber:
- CB9738829
- Molecular Formula:
- C23H38N6O6S
- Molecular Weight:
- 526.65
- MDL Number:
- MFCD00895735
- MOL File:
- 141396-28-3.mol
- MSDS File:
- SDS
| Melting point | 176-180° |
|---|---|
| alpha | D27 +76.1° (c = 1 in 0.2N HCl) |
| storage temp. | 2-8°C |
| solubility | DMSO: ≥20mg/mL |
| form | Solid |
| color | white to off-white |
| Merck | 14,779 |
| CAS DataBase Reference | 141396-28-3(CAS DataBase Reference) |
| FDA UNII | IY90U61Z3S |
| UNSPSC Code | 41116107 |
| NACRES | NA.77 |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS02 |
|---|---|
| Signal word | Warning |
| Hazard statements | H223+H229 |
| WGK Germany | 3 |
| RTECS | TM6126610 |
| HS Code | 29350090 |
| Toxicity | mouse,LD50,intraperitoneal,475mg/kg (475mg/kg),LUNGS, THORAX, OR RESPIRATION: DYSPNEABLOOD: NORMOCYTIC ANEMIABEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY),Yakuri to Chiryo. Pharmacology and Therapeutics. Vol. 14(Suppl, |
Argatroban price More Price(35)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich | A0487 | Argatroban monohydrate ≥98% (HPLC) | 141396-28-3 | 5mg | $84.1 | 2024-03-01 | Buy |
| Sigma-Aldrich | 1042408 | Argatroban monohydrate United States Pharmacopeia (USP) Reference Standard | 141396-28-3 | 350mg | $1560 | 2024-03-01 | Buy |
| TCI Chemical | A2705 | Argatroban Monohydrate >99.0%(HPLC) | 141396-28-3 | 25mg | $188 | 2024-03-01 | Buy |
| Sigma-Aldrich | A0487 | Argatroban monohydrate ≥98% (HPLC) | 141396-28-3 | 25mg | $340 | 2024-03-01 | Buy |
| TRC | A769005 | Argatroban monohydrate | 141396-28-3 | 25mg | $90 | 2021-12-16 | Buy |
Argatroban Chemical Properties,Uses,Production
Originator
Acova,SmithKline Beecham
Uses
Anticoagulant.
Uses
Argatroban monohydrate has been used as a thrombin-selective inhibitor in the thrombin assay of platelet poor plasma samples. It may be used as a standard in gradient reversed-phase liquid chromatography coupled with high-resolution multistage mass spectrometry (LC/HR-MSn) for photoproducts fragmentation analysis and as a thrombin inhibitor competitive assay with fullerene-based C60 nanoparticle (NPs).
Manufacturing Process
To a stirred solution of 28.3 g of NG-nitro-N2-(tert-butoxycarbonyl)-L-arginine
in 450 ml of dry tetrahydrofuran were added in turn 9.0 g of triethylamine
and 12.2 g of isobutyl chloroformate while keeping the temperature at -20°C.
After 10 minutes, to this was added 15.2 g of ethyl 4-methyl-2-
piperidinecarboxylate and the mixture was stirred for 10 minutes at -20°C. At
the end of this period, the reaction mixture was warmed to room
temperature. The solvent was evaporated and the residue taken up in 400 ml
of ethyl acetate, and washed successively with 200 ml of water, 100 ml of 5%
sodium bicarbonate solution, 100 ml of 10% citric acid solution and 200 ml of
water. The ethyl acetate solution was dried over anhydrous sodium sulfate.
The solution was evaporated to give 31.5 g (75 %) of ethyl 1-[NG-nitro-N2-
(tert-butoxycarbonyl)-L-arginyl]-4-methyl-2-piperidinecarboxylate in the form
of a syrup.
To a stirred solution of 30 g of ethyl 1-[NG-nitro-N2-(tert-butoxycarbonyl)-Larginyl]-
4-methyl-2-piperidinecarboxylate in 50 ml of ethyl acetate was added
80 ml of 10% dry HCl-ethyl acetate at 0°C. After 3 hours, to this solution was
added 200 ml of dry ethyl ether to precipitate a viscous oily product. This was
filtered and washed with dry ethyl ether to give ethyl 1-[NG-nitro-L-arginyl]-4-
methyl-2-piperidinecarboxylate hydrochloride as an amorphous solid.
To a stirred solution of ethyl 1-(NG-nitro-L-arginyl)-4-methyl-2-
piperidinecarboxylate hydrochloride in 200 ml of chloroform were added in
turn 18.5 g of triethylamine, and 14.7 g of 3-methyl-8-quinolinesulfonyl
chloride at 5°C, and stirring was continued for 3 hours at room temperature.
At the end of this period, the solution was washed twice with 50 ml of water.
The chloroform solution was dried over anhydrous sodium sulfate. Upon
evaporation of the solvent, the residue was chromatographed on 50 g of silica
gel packed in chloroform, washed with chloroform and eluted with 3%
methanol-chloroform. The fraction eluted from 3% methanol-chloroform was
evaporated to give 32.1 g (91%) of ethyl 1-[NG-nitro-N2-(3-methyl-8-
quinolinesulfonyl)-L-arginyl]-4-methyl-2-piperidinecarboxylate in the form of
an amorphous solid.
A solution of 30 g the above product in 100 ml of ethanol and 100 ml of 1 N
sodium hydroxide solution was stirred for 24 hrs at room temperature. At the
end of this period, the solution was neutralized with 1 N hydrochloric acid and
then concentrated to 70 ml. The solution was adjusted to pH=11 with 1 N
sodium hydroxide solution, washed three times with 100 ml of ethyl acetate,
acidified with 1 N hydrochloric acid and then extracted three times with 100
ml of chloroform. The combined chloroform solution was dried over anhydrous
sodium sulfate and evaporated to give 28.0 g (97%) of 1-[NG-nitro-N2-(3-
methyl-8-quinolinesulfonyl)-L-arginyl]-4-methyl-2-piperidinecarboxylic acid as
an amorphous solid. IR (KBr): 3,300, 1,720, 1,630 cm-1.
To a solution of 3.00 g of 1-[NG-nitro-N2-(3-methyl-8-quinolinesulfonyl)-Larginyl]-
4-methyl-2-piperidinecarboxylic acid in 50 ml of ethanol was added
0.5 g of palladium black and then the mixture was shaken under 10 kg/cm2
H2 pressure at 100°C for 8 hrs. At the end of this period, the ethanol solution
was filtered to remove the catalyst and evaporated to give 2.50 g (90%) of 1-
[N2-(3-methyl-1,2,3,4-tetrahydro-8-quinolinesulfonyl)-L-arginyl]-4-methyl-2-
piperidinecarboxylic acid as an amorphous solid. IR (KBr): 3,400, 1,620,
1,460, 1,380 cm-1.
brand name
Novastan (Mitsubishi Chemical Corporation, Japan).
Therapeutic Function
Anticoagulant
General Description
Argatroban anticoagulant is synthesized from L-arginine and photodegrades under light exposure.
Biochem/physiol Actions
Argatroban is a potent, seletive, univalent direct inhibitor of thrombin. It directly inhibits thrombin by binding only to its active site (thus univalent) as compared to Bivalent DTIs (hirudin and analogs) which bind both to the active site and exosite 1.
Enzyme inhibitor
This anticoagulant (FW = 508.64 g/mol; CAS 74863-84-6), also named (2R,4R) -4-methyl-1-N2-[ (3-methyl-1,2,3,4-tetrahydro-8-quinolilyl) sulfon- yl]-L-arginyl-2-piperidine carboxylic acid, Novastan?, and MD-805, is a direct thrombin inhibitor (1-11) that is an effective treatment for heparin- induced thrombocytopenia (HIT), an immunoglobulin-mediated adverse drug reaction characterized by platelet activation, thrombocytopenia, and a high risk of thrombotic complications among patients who are receiving or have recently received heparin. HIT is an autoimmune-like disorder, with the target antigen a multimolecular complex of the “self” protein, Platelet Factor-4, and heparin. Continued use of heparin is ill-advised and dangerous. Argatroban is also indicated for the treatment of heparin- induced thrombocytopenia and thrombosis syndrome (HITTS), an immune- mediated response to the administration of heparin that results in life- threatening thrombosis. Argatroban is one of four FDA-approved parenteral direct thrombin inhibitors, the others being lepirudin, desirudin, and bivalirudin. Argatroban is metabolized in the liver (t1/2 = ~50 min). Other DTI’s include recombinant hirudins, bivalirudin, and ximelagatran, either alone or in combination with melagatran. Argatroban is a significantly weaker inhibitor of trypsin, K = 4.3 μM, plasmin (K = ii 800 μM), coagulation factor Xa, and plasma kallikrein (Ki = 2 mM). Commercial sources often supply a mixture of 21 (R) and 21 (S) Diastereoisomers. The latter is more potent.
Argatroban Preparation Products And Raw materials
Raw materials
1of2
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | |
|---|---|---|---|---|---|
| Henan Bao Enluo International TradeCo.,LTD | +86-17331933971 +86-17331933971 | deasea125996@gmail.com | China | 2472 | 58 |
| BEIJING SJAR TECHNOLOGY DEVELOPMENT CO., LTD. | +86-18600796368 +86-18600796368 | sales@sjar-tech.com | China | 485 | 58 |
| Beijing Cooperate Pharmaceutical Co.,Ltd | 010-60279497 | sales01@cooperate-pharm.com | CHINA | 1803 | 55 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 | info@tianfuchem.com | China | 21630 | 55 |
| Shanghai Yingrui Biopharma Co., Ltd. | +86-21-33585366 - 03@ | sales03@shyrchem.com | CHINA | 738 | 60 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 | ivan@atkchemical.com | China | 33024 | 60 |
| career henan chemical co | +86-0371-86658258 +8613203830695 | sales@coreychem.com | China | 29862 | 58 |
| Xiamen AmoyChem Co., Ltd | +86-86-5926051114 +8615060885618 | sales@amoychem.com | China | 6383 | 58 |
| BOC Sciences | +1-631-485-4226 | inquiry@bocsci.com | United States | 19552 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 | sales@chemdad.com | China | 39894 | 58 |
View Lastest Price from Argatroban manufacturers
| Image | Update time | Product | Price | Min. Order | Purity | Supply Ability | Manufacturer | |
|---|---|---|---|---|---|---|---|---|
 |
2025-04-11 | Argatroban
141396-28-3
|
US $0.00 / g | 500g | 99%min | 100kgs | WUHAN FORTUNA CHEMICAL CO., LTD | |
 |
2025-04-04 | Argatroban
141396-28-3
|
US $0.00-0.00 / KG | 1KG | 98% | 1Ton | Henan Aochuang Chemical Co.,Ltd.
|
|
 |
2025-01-13 | Argatroban Monohydrate
141396-28-3
|
US $0.00 / g | 1g | More Than 99% | 100kg/Month | BEIJING SJAR TECHNOLOGY DEVELOPMENT CO., LTD. |
-

- Argatroban
141396-28-3
- US $0.00 / g
- 99%min
- WUHAN FORTUNA CHEMICAL CO., LTD
-

- Argatroban
141396-28-3
- US $0.00-0.00 / KG
- 98%
- Henan Aochuang Chemical Co.,Ltd.
-

- Argatroban Monohydrate
141396-28-3
- US $0.00 / g
- More Than 99%
- BEIJING SJAR TECHNOLOGY DEVELOPMENT CO., LTD.