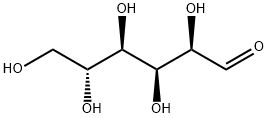
D(+)-Glucose
- CAS No.
- 50-99-7
- Chemical Name:
- D(+)-Glucose
- Synonyms
- D-GLUCOSE;DEXTROSE;D-Glucopyranose;Anhydrous Glucose;Hexose;D-(+)-GLUCOSE;(2R,3S,4R,5R)-2,3,4,5,6-Pentahydroxyhexanal;D-Glucose anhydrous;Glucose liquid;Emdex
- CBNumber:
- CB2250047
- Molecular Formula:
- C6H12O6
- Molecular Weight:
- 180.16
- MOL File:
- 50-99-7.mol
- Modify Date:
- 2024/9/5 14:45:50
| Melting point | 150-152 °C(lit.) |
|---|---|
| alpha | 52.75 º (c=10, H2O, NH4OH 25 ºC) |
| Boiling point | 232.96°C (rough estimate) |
| Density | 1.5440 |
| refractive index | 53 ° (C=10, H2O) |
| storage temp. | room temp |
| solubility | H2O: 1 M at 20 °C, clear, colorless |
| pka | pKa 12.43(H2O,t = 18,)(Approximate) |
| form | Crystalline Powder |
| color | White |
| PH | 5.0-7.0 (25℃, 1M in H2O) |
| Odor | Odorless |
| PH Range | 5.9 |
| optical activity | [α]25/D +52.5 to +53.0°(lit.) |
| Water Solubility | Soluble |
| λmax |
λ: 260 nm Amax: 0.03 λ: 280 nm Amax: 0.02 |
| Merck | 14,4459 |
| BRN | 1281608 |
| Stability | Stable. Substances to be avoided include strong oxidizing agents. Combustible. |
| InChIKey | WQZGKKKJIJFFOK-DVKNGEFBSA-N |
| LogP | -2.490 (est) |
| CAS DataBase Reference | 50-99-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Glucose(50-99-7) |
| EPA Substance Registry System | Dextrose (50-99-7) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS07 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Warning | |||||||||
| Hazard statements | H302-H227 | |||||||||
| Precautionary statements | P501-P270-P210-P264-P280-P370+P378-P301+P312+P330-P403+P235 | |||||||||
| Hazard Codes | Xi,Xn | |||||||||
| Risk Statements | 36/37/38-63-62-46-36/38-21 | |||||||||
| Safety Statements | 26-36/37-24/25-53-25 | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | LZ6600000 | |||||||||
| F | 3 | |||||||||
| Autoignition Temperature | 500 °C | |||||||||
| TSCA | Yes | |||||||||
| HS Code | 17023051 | |||||||||
| Toxicity | LD50 orally in Rabbit: 25800 mg/kg | |||||||||
| NFPA 704 |
|
D(+)-Glucose price More Price(51)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | RDD016 | D-(+)-Glucose anhydrous, free-flowing, Redi-Dri?, ≥99.5% | 50-99-7 | 500G | ₹5958.4 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | RDD016 | D-(+)-Glucose anhydrous, free-flowing, Redi-Dri?, ≥99.5% | 50-99-7 | 1KG | ₹6889.4 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | RDD016 | D-(+)-Glucose anhydrous, free-flowing, Redi-Dri?, ≥99.5% | 50-99-7 | 2.5KG | ₹8113 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | RDD016 | D-(+)-Glucose anhydrous, free-flowing, Redi-Dri?, ≥99.5% | 50-99-7 | 12KG | ₹23155.3 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | G8270 | D-(+)-Glucose ≥99.5% (GC) | 50-99-7 | 1KG | ₹3430 | 2022-06-14 | Buy |
D(+)-Glucose Chemical Properties,Uses,Production
Description
D(+)-Glucose is one of the most important biological compounds found in nature. It is a main product in photosynthesis and is oxidized in cellular respiration. D(+)-Glucose polymerizes to form several important classes of biomolecules including cellulose, starch, and glycogen. It also combines with other compounds to produce common sugars such as sucrose and lactose. The form of D(+)-Glucose displayed above is D-D(+)-Glucose. The “D” designation indicates the configuration of the molecule. The “D” configuration specifies that the hydroxyl group on the number 5 carbon is on the right side of the molecule. The mirror image of D-D(+)-Glucose produces another form of D(+)-Glucose called L-D(+)-Glucose.D(+)-Glucose is the most common form of a large class of molecules called carbohydrates. Carbohydrates are the predominant type of organic compounds found in organisms and include sugar, starches, and fats. Carbohydrates, as the name implies, derive their name from D(+)-Glucose,C6H12O6, which was considered a hydrate of carbon with the general formula of Cn(H2O)n, where n is a positive integer. Although the idea of water bonded to carbon to form a hydrate of carbon was wrong, the term carbohydrate persisted. Carbohydrates consist of carbon, hydrogen, and oxygen atoms, with the carbon atoms generally forming long unbranched chains. Carbohydrates are also known as saccharides derived from the Latin word for sugar, saccharon.
Chemical Properties
White or almost white, crystalline powder.
History
D(+)-Glucose is the most important and predominant monosaccharide found in nature. It was isolated from raisins by Andreas Sigismund Marggraf (1709–1782) in 1747, and in 1838, Jean-Baptiste-André Dumas (1800–1884) adopted the name glucose from the Greek word glycos meaning sweet. Emil Fischer (1852–1919) determined the structure of glucose in the late 19th century. Glucose also goes by the names dextrose (from its ability to rotate polarized light to the right), grape sugar, and blood sugar. The term blood sugar indicates that glucose is the primary sugar dissolved in blood. Glucose’s abundant hydroxyl groups enable extensive hydrogen bonding, and so glucose is highly soluble in water.
Uses
D(+)-Glucose anhydrous for biochemistry Reag. Ph Eur. CAS 50-99-7, molar mass 180.16?g/mol.
Definition
ChEBI: The open chain form of D-glucose.
General Description
Watery odorless colorless liquid. Denser than water and soluble in water. Hence sinks in and mixes with water.
Air & Water Reactions
Water soluble.
Reactivity Profile
A weak reducing agent.
Health Hazard
No toxicity
Safety Profile
Mildly toxic by ingest ion. An experimental teratogen. Experi mental reproductive effects. Questionable carcinogen with experimental tumorigenic data. Mutation data reported. Potentially explosive reaction with potassium nitrate + sodium peroxide when heated in a sealed container. Uxtures with alkali release carbon monoxide when heated. When heated to decomposition it emits acrid smoke and irritating fumes.
Purification Methods
Crystallise -D-glucose from hot glacial acetic acid or pyridine. Traces of solvent are removed by drying in a vacuum oven at 75o for >3hours. [Gottfried Adv Carbohydr Chem 5 127 1950, Kjaer & Lindberg Acta Chem Scand 1 3 1713 1959, Whistler & Miller Methods in Carbohydrate Chemistry I 1301962, Academic Press, Beilstein 1 IV 4306.] [For equilibrium forms see Angyal Adv Carbohydr Chem 42 15 1984, Angyal & Pickles Aust J Chem 25 1711 1972.]
D(+)-Glucose Preparation Products And Raw materials
Raw materials
Preparation Products
1of8
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Vardhaman P Golechha | 9704422000 | Telangana, India | 294 | 58 | Inquiry |
| Sudarshan Pharma Industries Limited | +91-2242221111 +91-9320932107 | Maharashtra, India | 37 | 58 | Inquiry |
| ADVENT CHEMBIO PRIVATE LIMITED | +91-9821524116 +91-9821524116 | Mumbai, India | 353 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| HRV Global Life Sciences | +91-9820219686 +91-9820219686 | Telangana, India | 379 | 58 | Inquiry |
| Sujata Chemicals | +91-9898025496 +91-9898025496 | Gujarat, India | 73 | 58 | Inquiry |
| Sayaji Maize Products (Sayaji Group) | +91-22901585 +91-9898032040 | Gujarat, India | 6 | 58 | Inquiry |
| Nandu Chemical Industries | +91-8362330469 +91-8362330469 | Karnataka, India | 84 | 58 | Inquiry |
| Rajaram And Brothers | +91-9425772222 +91-9926772222 | Madhya Pradesh, India | 7 | 58 | Inquiry |
| Rubexco Pvt Ltd | +91-9825231936 +91-9909917613 | Gujarat, India | 36 | 58 | Inquiry |
Related articles
- Application of D(+)-Glucose in various fields
- D(+)-Glucose is a monosaccharide belonging to the group of carbohydrates, a very important biological compound that makes up t....
- Aug 19,2024
- Is glucose a polar molecule?
- Glucose is a polar molecule due to its multiple hydroxyl groups, which create polar bonds. The polarity of glucose plays a ess....
- Dec 20,2023
- D-Glucose VS L-Glucose
- The passage introduces the differences between D-Glucose and L-Glucose.
- Nov 25,2022
50-99-7(D(+)-Glucose)Related Search:
1of4
chevron_right




