ChemicalBook > Product Catalog >Analytical Chemistry >Standard >Pharmaceutical Impurity Reference Standards >SalMeterol Xinafoate Adduct IMpurity
SalMeterol Xinafoate Adduct IMpurity
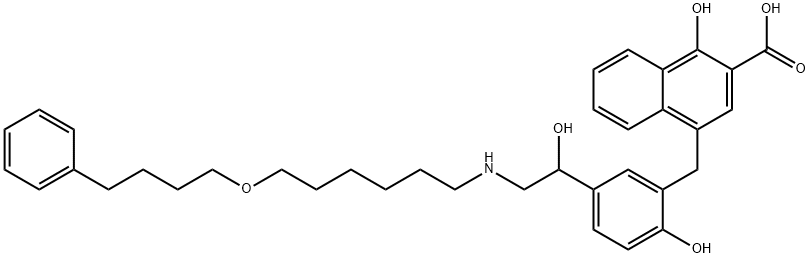
- CAS No.
- 1330076-52-2
- Chemical Name:
- SalMeterol Xinafoate Adduct IMpurity
- Synonyms
- Salmeterol USP RC H;SalMeterol IMpurity H;Salmeterol Impurity 14;Salmeterol EP Impurity H;salmeterol impurities1071;SalMeterol Related CoMpound H;Salmeterol USP Related Compound H;SalMeterol Xinafoate Adduct IMpurity;Salmeterol Impurity (Xinafoate Adduct Impurity);SalMeterol Xinafoate Adduct IMpurity DISCONTINUED
- CBNumber:
- CB32545735
- Molecular Formula:
- C36H43NO6
- Molecular Weight:
- 585.73
- MOL File:
- 1330076-52-2.mol
- MSDS File:
- SDS
- Modify Date:
- 2023/8/16 18:05:29
SalMeterol Xinafoate Adduct IMpurity Preparation Products And Raw materials
Raw materials
Preparation Products
Global( 50)Suppliers
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-87569266 15319487004 | China | 4088 | 58 | Inquiry |
| Hebei Yanxi Chemical Co., Ltd. | +86-17531190177; +8617531190177 | China | 5873 | 58 | Inquiry |
| Career Henan Chemica Co | +86-0371-86658258 +8613203830695 | China | 30253 | 58 | Inquiry |
| Baoji Guokang Healthchem co.,ltd | +8615604608665 15604608665 | CHINA | 9427 | 58 | Inquiry |
| sgtlifesciences pvt ltd | +8617013299288 | China | 12382 | 58 | Inquiry |
| China National Standard Pharmaceutical Corporation Limited | +8615391658522 | China | 11927 | 58 | Inquiry |
| HUNAN CHEMFISH SCIENTIFIC CO.,LTD | +86-0731-85567275 13021512891 | China | 7012 | 58 | Inquiry |
1330076-52-2(SalMeterol Xinafoate Adduct IMpurity)Related Search:
Salmeterol EP Impurity A
Salmeterol Impurity 9
Salmeterol
SalMeterol EP IMpurity G
Benzenebutanol, 4-bromo-
3-Bromo-benzenebutanol
Salmeterol xinafoate
4-Hydroxy-α1-[[[6-(2-phenylethoxy)hexyl]aMino]Methyl]-1,3-benzenediMethanol
Salbutamol Impurity 25
SalMeterol EP IMpurity F
chevron_left
1of4
chevron_right
1-Hydroxy-4-[[2-hydroxy-5-[1-hydroxy-2-[[6-(4-phenylbutoxy)hexyl]amino]ethyl]phenyl]methyl]-2-naphthalenecarboxylic acid
1-Hydroxy-4-[2-hydroxy-5-(1-hydroxy-2-{[6-(4-phenylbutoxy)hexyl]amino}ethyl)benzyl]-2-naphthoic acid
Salmeterol Related Compound H (15 mg) (1-hydroxy-4-[2-hydroxy-5-(1-hydroxy-2-{[6-(4-phenylbutoxy)hexyl]amino}ethyl)benzyl]-2-naphthoic acid)
Salmeterol USP RC H
SalMeterol Xinafoate Adduct IMpurity
SalMeterol Xinafoate Adduct IMpurity DISCONTINUED
SalMeterol Related CoMpound H
1-Hydroxy-4-(2-hydroxy-5-{1-hydroxy-2-[6-(4-phenylbutoxy)hexylaMino]ethyl}benzyl)naphthalene-2-carboxylic Acid
SalMeterol IMpurity H
Salmeterol Impurity 9(Salmeterol Related Compound H)
1-hydroxy-4-[[2-hydroxy-5-[1-hydroxy-2-[6-(4-phenylbutoxy)hexylamino]ethyl]phenyl]methyl]naphthalene-2-carboxylic acid
Salmeterol Impurity 14
Salmeterol Impurity H
(Xinafoate Adduct Impurity)
Salmeterol USP Related Compound H
2-Naphthalenecarboxylic acid, 1-hydroxy-4-[[2-hydroxy-5-[1-hydroxy-2-[[6-(4-phenylbutoxy)hexyl]amino]ethyl]phenyl]methyl]-
Salmeterol Impurity 14 (Xinafoate Adduct Impurity)
Salmeterol Related Compound H (Salmeterol Xinafoate Adduct Impurity)
salmeterol impurities1071
Salmeterol Related Compound H (1-hydroxy-4-[2-hydroxy-5-(1-hydroxy-2-{[6-(4-phenylbutoxy)hexyl]amin (1609680)
Salmeterol Impurity 13 (Xinafoate Adduct Impurity)
Salmeterol EP Impurity H
Salmeterol Impurity (Xinafoate Adduct Impurity)
1330076-52-2
Amines
Aromatics
Intermediates & Fine Chemicals
Pharmaceuticals
Impurity





