Tafamidis Meglumine
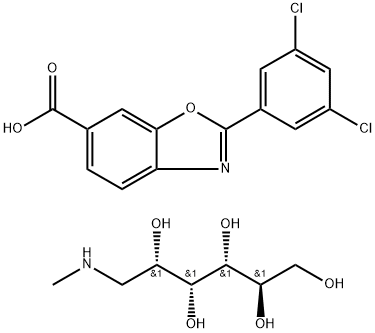
- CAS No.
- 951395-08-7
- Chemical Name:
- Tafamidis Meglumine
- Synonyms
- PF06291826;PF-06291826;cas 594839-88-0;afamidis Meglumine;Tafamidis Meglumine;Tafamidis-meglumine API;He prescribed metoclopramide;Tafamidis meglumine (Fx1006, PF06291826);tafamidis meglumine,Inhibitor,Tafamidis,inhibit;TAFAMIDIS;CAS 594839-88-0;PF-06291826;PF06291826
- CBNumber:
- CB53159986
- Molecular Formula:
- C21H24Cl2N2O8
- Molecular Weight:
- 503.33
- MOL File:
- 951395-08-7.mol
- Modify Date:
- 2024/8/28 16:27:42
| Melting point | 195 - 198°C |
|---|---|
| storage temp. | -20°C Freezer, Under inert atmosphere |
| solubility | DMSO (Sparingly, Heated, Sonicated) |
| form | Solid |
| color | White to Off-White |
| InChIKey | DQJDBUPLRMRBAB-KUAVVOKVNA-N |
| SMILES | [C@@H](O)([C@@H](O)CNC)[C@H](O)[C@H](O)CO.ClC1=CC(Cl)=CC(C2=NC3=CC=C(C(=O)O)C=C3O2)=C1 |&1:0,2,7,9,r| |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |  GHS08 |
|---|---|
| Signal word | Danger |
| Hazard statements | H360 |
| Precautionary statements | P201-P202-P281-P308+P313-P405-P501 |
Tafamidis Meglumine Chemical Properties,Uses,Production
Description
In November 2011, the European Commission approved tafamidis meglumine (Fx-1006A, PF-06291826) for the treatment of transthyretin familial amyloid polyneuropathy (TTR-FAP) in adult patients with stage 1 symptomatic polyneuropathy. Tafamidis stabilizes both the wild type and mutant forms of TTR tetramer and prevents tetramer dissociation by noncooperatively binding to the two thyroxine binding sites. Tafamidis is the first approved medicine for TTR-FAP. The Kd values for tafamidis for the two thyroxine binding sites on TTR, as determined by isothermal titration calorimetry, were 3 nM and 278 nM, respectively. In another in vitro study using wild type TTR, V30M mutant TTR, and V122I mutant TTR, it was shown that tafamidis inhibited fibril formation in a concentration-dependent manner reaching EC50 at a tafamidis:TTR stoichiometry of <1 (EC50 was in the range of 2.7–3.2 μM, corresponding to a tafamidis:TTR stoichiometry range of 0.75–0.9). Tafamidis has been synthesized by coupling 4-amino-3- hydroxybenzoic acid with 3,5-dichlorobenzoyl chloride followed by dehydration using p-toluenesulfonic acid.
Uses
Tafamidis Meglumine is used as a potential therapeutics for COVID-19 and related viral infections
Clinical Use
Tafamidis meglumine is a transthyretin amyloid inhibitor that was approved for the treatment of transthyretin amyloid polyneuropathy (ATTR-PN) and transthyretin familial amyloid polyneropathy (TTR-FAP). These diseases represent a rare autosomal neurodegenerative disorder characterized by autonomic, sensory and motor impairment which are typically fatal. Tafamidis was discovered at The Scripps Research Institute and developed by Pfizer.
Tafamidis Meglumine Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Dr. Reddy's Laboratories Ltd | +91-4049002900 +91-4049002900 | Hyderabad, India | 165 | 58 | Inquiry |
| CVR Life sciences Pvt Ltd | +91-7386789224 +91-9912960099 | Hyderabad, India | 22 | 58 | Inquiry |
| Synaptics Labs Private Limited | +91-8341963666 +91-8341963666 | Hyderabad, India | 84 | 58 | Inquiry |
| Nuray Chemicals Pvt Ltd | +91-4066616700 +91-8220444745 | Tamil Nadu, India | 49 | 58 | Inquiry |
| Aruvi Labs | +91-8056181939 +91-8056181939 | Tamil Nadu, India | 149 | 58 | Inquiry |
| Biophore India Pharmaceuticals Pvt Ltd | +91-9100031592 +91-9030907714 | Telangana, India | 134 | 58 | Inquiry |
| Morepen Laboratories Ltd | +91-7017029282 +91-9818305894 | Haryana, India | 34 | 58 | Inquiry |
| MSN LABORATORIES PRIVATE LTD | +91 40 30438600 | New Delhi, India | 230 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| Pharmaffiliates Analytics and Synthetics P. Ltd | +91-172-5066494 | Haryana, India | 6773 | 58 | Inquiry |





