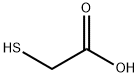
Thioglycolic acid
- CAS No.
- 68-11-1
- Chemical Name:
- Thioglycolic acid
- Synonyms
- THIOGLYCOLIC ACID;TGA;2-MERCAPTOACETIC ACID;MEQUINDOX;thioglycolic;Thioglycolate;THIOGLYCOLLIC ACID;Mercaptoacetate;2-mercaptoacetate;mercaptoacetic
- CBNumber:
- CB6477604
- Molecular Formula:
- C2H4O2S
- Molecular Weight:
- 92.12
- MOL File:
- 68-11-1.mol
- MSDS File:
- SDS
- Modify Date:
- 2024/8/21 22:41:43
| Melting point | −16 °C(lit.) |
|---|---|
| Boiling point | 96 °C5 mm Hg(lit.) |
| Density | 1.326 g/mL at 20 °C(lit.) |
| vapor density | 3.2 (vs air) |
| vapor pressure | 0.4 mm Hg ( 25 °C) |
| refractive index |
n |
| Flash point | 126 °C |
| storage temp. | Store at +2°C to +8°C. |
| solubility | Chloroform (Sparingly), Methanol (Sparingly) |
| form | Liquid |
| pka | 3.68(at 25℃) |
| color | clear clear, colorless |
| PH | 1 (H2O, 20℃) |
| Odor | strong unpleasant odor |
| Water Solubility | soluble |
| Sensitive | Air Sensitive |
| Merck | 14,9336 |
| BRN | 506166 |
| Exposure limits | TLV-TWA 1 ppm (~3.8 mg/m3) (ACGIH). |
| Stability | Air Sensitive |
| InChIKey | CWERGRDVMFNCDR-UHFFFAOYSA-N |
| LogP | 0.090 |
| CAS DataBase Reference | 68-11-1(CAS DataBase Reference) |
| NIST Chemistry Reference | Acetic acid, mercapto-(68-11-1) |
| EPA Substance Registry System | Thioglycolic acid (68-11-1) |
SAFETY
Risk and Safety Statements
| Symbol(GHS) |   GHS05,GHS06 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H301+H311+H331-H314-H317 | |||||||||
| Precautionary statements | P261-P270-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338 | |||||||||
| Hazard Codes | T,T+ | |||||||||
| Risk Statements | 23/24/25-34-26-24/25 | |||||||||
| Safety Statements | 25-27-28-45-36/37-28C-23 | |||||||||
| RIDADR | UN 1940 8/PG 2 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 1 ppm (4 mg/m3) [skin] | |||||||||
| WGK Germany | 1 | |||||||||
| RTECS | AI5950000 | |||||||||
| F | 9-13-23 | |||||||||
| Autoignition Temperature | 662 °F | |||||||||
| TSCA | Yes | |||||||||
| HazardClass | 8 | |||||||||
| PackingGroup | II | |||||||||
| HS Code | 29309070 | |||||||||
| Toxicity | LD50 orally in rats: 0.15 ml/kg (Deichmann, Mergard) | |||||||||
| NFPA 704 |
|
Thioglycolic acid price More Price(28)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | T6750 | Thioglycolic acid solution ~70?% (w/w) in H2O | 68-11-1 | 50ML | ₹2662.95 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | T6750 | Thioglycolic acid solution ~70?% (w/w) in H2O | 68-11-1 | 500ML | ₹11561.1 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | T3758 | Thioglycolic acid 98% | 68-11-1 | 100ML | ₹4806.3 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | T3758 | Thioglycolic acid 98% | 68-11-1 | 500ML | ₹8194.53 | 2022-06-14 | Buy |
| Sigma-Aldrich(India) | 88652 | Thioglycolic acid solution ~80% in H2O, for spectrophotometric det. of palladium, iron, uranium(VI), molybdates and nitrites | 68-11-1 | 250ML | ₹3669.68 | 2022-06-14 | Buy |
Thioglycolic acid Chemical Properties,Uses,Production
Description
Thio glycolic acid (TGA) is the organic compound HSCH2CO2H . It contains both a thiol (mercaptan) and a carboxylic acid. It is a clear liquid with a strong unpleasant odor. It is readily oxidized by air to the corresponding disulfide [SCH2CO2H]2.
TGA was developed in the 1940s for use as a chemical depilatory and is still used as such, especially in salt forms, including calcium thioglycolate and sodium thioglycolate. TGA is the precursor to ammonium thioglycolate that is used for permanents. TGA and its derivatives break the disulfide bonds in the cortex of hair. One reforms these broken bonds in giving hair a "perm." Alternatively and more commonly, the process leads to depilation as is done commonly in leather processing. It is also used as an acidity indicator, manufacturing of thioglycolates, and in bacteriology for preparation of thioglycolate media.
TGA is also used in the making of tin stabilizers often used in certain polyvinyl chloride products (such as vinyl siding).
TGA, usually as its dianion, forms complexes with metal ions. Such complexes have been used for the detection of iron, molybdenum, silver, and tin.
Thioglycolic acid is used as nucleophile in thioglycolysis reactions used on condensed tannins to study their structure.
Chemical Properties
Also known as mercaptoacetic acid, HSCH2COOH is a colorless liquid with a strong unpleasant odor. Used as a reagent for metals such as iron, molybdenum, silver, and tin,and in bacteriology.
Uses
Mercaptoacetic acid is used as a reagent formetals analysis; in the manufacture of thioglycolates, pharmaceuticals, and permanentwave solutions; and as a vinyl stabilizer.
Preparation
Thioglycolic acid is prepared from Sodium sulfide plus Sodium monochloride acetate to yield Dithioglycollic acid. Electrolysis of the latter acid yields the title material.
General Description
A colorless liquid with an unpleasant odor. Density 1.325 g / cm3. Used to make permanent wave solutions and depilatories. Corrosive to metals and tissue.
Air & Water Reactions
Readily oxidized by air. Water soluble.
Reactivity Profile
Mercaptoacetic acid is readily oxidized by air . Reacts readily with other oxidizing agents as well in reactions that may generate toxic gases. Incompatible with diazo and azo compounds, halocarbons, isocyanates, aldehydes, alkali metals, nitrides, hydrides, and other strong reducing agents. Reactions with these materials may generate heat and toxic and flammable gases. May react with acids to liberate hydrogen sulfide. Neutralizes bases in exothermic reactions. Reacts with cyanides, sulfites, nitrites, thiosulfates to generate flammable and toxic gases and heat. Reacts with carbonates and bicarbonates.
Hazard
Toxic by ingestion and inhalation, strong irritant to tissue, eyes, and skin.
Health Hazard
Mercaptoacetic acid is a highly toxic and ablistering compound. Even a 10% solutionwas lethal to most experimental animals by dermal absorption. The oral LD50 value ofundiluted acid is less than 50 mg/kg (Patty1963). The lethal dose in rabbits by skinabsorption is 300 mg/kg. The acute toxicsymptoms in test animals include weakness,respiratory distress, convulsions, irritation ofthe gastrointestinal tract, and liver damage.Mercaptoacetic acid is a severe irritant.Contact with eyes can cause conjunctivalinflammation and corneal opacity. Skin contact can result in burns and necrosis.
Fire Hazard
Combustible material: may burn but does not ignite readily. When heated, vapors may form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated. Runoff may pollute waterways. Substance may be transported in a molten form.
Safety Profile
Poison by ingestion, skin contact, intraperitoneal, and intravenous routes. Moderately toxic by subcutaneous route. A corrosive irritant to skin, eyes, and mucous membranes. When heated to decomposition it emits toxic fumes of SOx. See also MERCAPTANS and HYDROGEN SULFIDE.
Potential Exposure
Thioglycolic acid is used to make thioglycolates; in sensitivity tests for iron; in formulations of permanent wave solutions and depilatories; in pharmaceutical manufacture; as a stabilizer in vinyl plastics.
Carcinogenicity
Thioglycolic acid was not mutagenic in a number of Salmonella typhimurium strains with or without metabolic activation.
Shipping
UN1940 Thyoglycolic acid, Hazard class: 8; Labels: 8-Corrosive material.
Purification Methods
Mix the acid with an equal volume of *benzene; the *benzene is then distilled off to dehydrate the acid. After heating to 100o to remove most of the *benzene, the residue is distilled under vacuum and stored in sealed ampoules at 3o. [Eshelman et al. Anal Chem 22 844 1960, Beilstein 3 IV 1130.]
Incompatibilities
Compounds of the carboxyl group react with all bases, both inorganic and organic (i.e., amines) releasing substantial heat, water and a salt that may be harmful. Incompatible with arsenic compounds (releases hydrogen cyanide gas), diazo compounds, dithiocarbamates, isocyanates, mercaptans, nitrides, and sulfides (releasing heat, toxic and possibly flammable gases), thiosulfates and dithionites (releasing hydrogen sulfate and oxides of sulfur). Air, strong oxidizers; bases, active metals, for example, sodium potassium, magnesium, and calcium. Readily oxidized by air. Thermal decomposition causes release of hydrogen sulfide. May attack various metals.
Waste Disposal
Dissolve in flammable solvent and burn in furnace equipped with afterburner and alkaline scrubber.
Thioglycolic acid Preparation Products And Raw materials
Raw materials
1of3
chevron_rightPreparation Products
1of3
chevron_right| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| ANJI BIOSCIENCES | +91-9000100077 +91-9000100077 | Hyderabad, India | 430 | 58 | Inquiry |
| ULTIMA CHEMICALS | +91 7208029458 | Mumbai, India | 95 | 58 | Inquiry |
| JSK Chemicals | +919879767970 | Gujarat, India | 3756 | 58 | Inquiry |
| Titan Biotech Limited | 09711169006 | Delhi, India | 196 | 58 | Inquiry |
| Aim Chemicals & Ingredients | 91-22-66252000 | Maharashtra, India | 80 | 58 | Inquiry |
| Molychem | 08048372666Ext 559 | Mumbai, India | 577 | 58 | Inquiry |
| Amrit Chem | 08048987434 | Delhi, India | 73 | 58 | Inquiry |
| KIP Chemicals Pvt., Ltd. | 91-22-49730577 | Maharashtra, India | 4 | 58 | Inquiry |
| S.R. Drugs And Intermediates Pvt. Ltd. | +91-08455-244489 | Telangana, India | 3 | 58 | Inquiry |
| Oxford laboratories | +91 250 239 0032 | Maharashtra, India | 652 | 58 | Inquiry |
| Supplier | Advantage |
|---|---|
| ANJI BIOSCIENCES | 58 |
| ULTIMA CHEMICALS | 58 |
| JSK Chemicals | 58 |
| Titan Biotech Limited | 58 |
| Aim Chemicals & Ingredients | 58 |
| Molychem | 58 |
| Amrit Chem | 58 |
| KIP Chemicals Pvt., Ltd. | 58 |
| S.R. Drugs And Intermediates Pvt. Ltd. | 58 |
| Oxford laboratories | 58 |
Related articles
- Different applications of thioglycolic acid
- Thioglycolic acid is an organic acid with the chemical formula C2H4O2S. Toxic, colorless and transparent liquid with strong pu....
- Apr 20,2022
Related Qustion
- Q:What is thioglycolic acid used for skin?
- A:TGA (active ingredient: 2-mercaptoacetic acid) causes depigmentation in spots of ferric origin. Unlike chelators, like EDTA an....
- Jun 17,2024





