5-(p-isocyanatobenzyl)-o-tolyl isocyanate
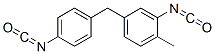
- CAS No.
- 75790-84-0
- Chemical Name:
- 5-(p-isocyanatobenzyl)-o-tolyl isocyanate
- Synonyms
- 5-(p-isocyanatobenzyl)-o-tolyl isocyanate;BENZENE,2-ISOCYANATO-4-((4-ISOCYANATOPHENYL)METHYL)-1-MET.;2-Isocyanato-4-[(4-isocyanatophenyl)methyl]-1-methylbenzene
- CBNumber:
- CB6938073
- Molecular Formula:
- C16H12N2O2
- Molecular Weight:
- 264.27868
- MOL File:
- 75790-84-0.mol
| EPA Substance Registry System | 4-Methyldiphenylmethane-3,4-diisocyanate (75790-84-0) |
|---|
5-(p-isocyanatobenzyl)-o-tolyl isocyanate Chemical Properties,Uses,Production
Uses
5-?(p-?Isocyanatobenzyl)?-?O-?tolyl Isocyanate is used in the manufacture of polyurethanes.Environmental toxin on US EPA Toxic Release Inventory list (TRI) list.
Reactivity Profile
Isocyanates and thioisocyanates, such as 5-(p-isocyanatobenzyl)-o-tolyl isocyanate, are incompatible with many classes of compounds, reacting exothermically to release toxic gases. Reactions with amines, aldehydes, alcohols, alkali metals, ketones, mercaptans, strong oxidizers, hydrides, phenols, and peroxides can cause vigorous releases of heat. Acids and bases initiate polymerization reactions in these materials. Some isocyanates react with water to form amines and liberate carbon dioxide. Base-catalysed reactions of isocyanates with alcohols should be carried out in inert solvents. Such reactions in the absence of solvents often occur with explosive violence, [Wischmeyer(1969)].





