Tapentadol Hydrochloride
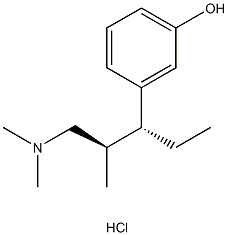
- CAS No.
- 175591-09-0
- Chemical Name:
- Tapentadol Hydrochloride
- Synonyms
- Tapentediol;Tapentadol hcl;Tapentadol Hydrochloride;Tapentadol (hydrochloride) (CRM);Tapentadol hydrochloride solution;Tapentadol hydrochloride(Y0002120);3-[(1R,2R)-3-(DiMethylaMino)-1-ethyl-2-Methylpro;Tapentadol Hydrochloride (1.0 mg/mL in Methanol);3-((2R,3R)-1-(diMethylaMino)-2-Methylpentan-3-yl)phenol hydrochloride;3-[(1R,2R)-3-(Dimethylamino)-1-ethyl-2-methylpropyl]phenol Hydrochloride
- CBNumber:
- CB71565532
- Molecular Formula:
- C14H24ClNO
- Molecular Weight:
- 257.81
- MOL File:
- 175591-09-0.mol
- Modify Date:
- 2023/11/2 15:05:42
SAFETY
Risk and Safety Statements
| Symbol(GHS) |    GHS02,GHS06,GHS08 |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Signal word | Danger | |||||||||
| Hazard statements | H225-H301+H311+H331-H370 | |||||||||
| Precautionary statements | P210-P260-P280-P301+P310-P311 | |||||||||
| Hazard Codes | F,T | |||||||||
| Risk Statements | 11-23/24/25-39/23/24/25 | |||||||||
| Safety Statements | 7-16-36/37-45 | |||||||||
| RIDADR | UN1230 - class 3 - PG 2 - Methanol, solution | |||||||||
| WGK Germany | 1 | |||||||||
| NFPA 704 |
|
Tapentadol Hydrochloride price More Price(1)
| Manufacturer | Product number | Product description | CAS number | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|---|
| Sigma-Aldrich(India) | T-058 | Tapentadol hydrochloride solution 1.0?mg/mL in methanol (as free base), ampule of 1?mL, certified reference material, Cerilliant? | 175591-09-0 | 1ML | ₹22184.4 | 2022-06-14 | Buy |
| Product number | Packaging | Price | Buy |
|---|---|---|---|
| T-058 | 1ML | ₹22184.4 | Buy |
Tapentadol Hydrochloride Chemical Properties,Uses,Production
Description
Agonism of the MOR is a common strategy for moderate to severe pain intervention. Opioid drugs, such as morphine, that modulate this receptor have demonstrated efficacy in acute situations; however, chronic conditions, particularly those of neuropathic or inflammatory etiology, suffer from inadequate pain management with this treatment. With a narrow therapeutic window, traditional MOR agonists flirt with side effects at optimal analgesia, and prolonged use increases the potential for physical dependency. Since extensive efforts to design activators of MOR have failed to dissociate the undesirable adverse effects from the analgesic properties, the focus has been on enhancing the analgesic efficacy through a dual mechanism of action. Tapentadol hydrochloride brings this concept to fruition; MOR agonism is coupled with noradrenaline reuptake inhibition in a combinatory contribution to analgesia. Compared to morphine, it is about 50-fold less potent for MOR (Ki = 100 nM for tapentadol versus 2 nM for morphine). The Ki for inhibition of noradrenaline reuptake was 500 nM while reuptake of serotonin was only weakly inhibited (Ki = 2.5μM). Despite its lower affinity for MOR, the dual mechanism has provided an efficacious profile in both acute and chronic conditions with fewer side effects.
Chemical Properties
Light Brown Solid
Uses
A novel, centrally acting oral analgesic with a dual mode of action that has demonstrated efficacy in preclinical and clinical models of pain relief.
Clinical Use
Tapentadol was approved by the FDA in November 2008 for the treatment of moderate to severe acute pain. It is a centrally acting analgesic that acts as both an agonist at the l-opiod receptor and as a norepinephrine re-uptake inhibitor, allowing it to have efficacy similar to potent narcotic analgesics but without their side effects. The drug was developed by Grunenthal and Johnson & Johnson and was marketed starting in 2009.
Side effects
The common adverse effects of tapentadol were nausea, vomiting, somnolence, dizziness, and itching. As with all opioid medications, constipation was also an issue. Tapentadol is contraindicated in patients taking monoamine oxidase inhibitors because of the potential for adverse cardiovascular events due to additive effects on norepinephrine levels. As with other MOR agonists, it is also contraindicated in patients with paralytic ileus. In patients with a history of epilepsy or seizure, tapentadol may induce seizures. Since tapentadol causes somnolence, its combination with other sleep aids could dangerously affect breathing. Similarly, patients with existing breathing or lung problems are cautioned about using tapentadol. Furthermore, patients with past or present substance abuse or drug addiction should consult the doctor prior to use since physical dependency and addiction is a risk with tapentadol. Alcohol should be avoided due to the potential additive effect on CNS depression.
Tapentadol Hydrochloride Preparation Products And Raw materials
Raw materials
Preparation Products
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| Micro Orgo Chem | +91-91-022-24969510 +91-9082984052 | Mumbai, India | 66 | 58 | Inquiry |
| AARTIA KEM SCIENCE | +91-8291072530 +91-8291072530 | Maharashtra, India | 70 | 58 | Inquiry |
| Varanous Labs Pvt Ltd | +91-7036248882 +91-7036248882 | Hyderabad, India | 1541 | 58 | Inquiry |
| Synaptics Labs Private Limited | +91-8341963666 +91-8341963666 | Hyderabad, India | 84 | 58 | Inquiry |
| Viyash Life Sciences Pvt Ltd | +91-4023635052 +91-4023635052 | Telangana, India | 213 | 58 | Inquiry |
| Chemeca Drugs Private Limited (Vegesna Laboratories Pvt Ltd) | +91-8924236065 +91-9951616502 | AndhraPradesh, India | 48 | 58 | Inquiry |
| Virupaksha Organics Pvt Ltd | +91-9440049755 +91-9848648601 | Telangana, India | 40 | 58 | Inquiry |
| Precise Biopharma Pvt Ltd | +91-2267828600 +91-2267828600 | Maharashtra, India | 46 | 58 | Inquiry |
| Amara Labs Pvt Ltd | +91-9553279972 +91-9100091565 | Hyderabad, India | 42 | 58 | Inquiry |
| Arene Lifesciences Limited | +91-9849847907 +91-8455241148 | Telangana, India | 55 | 58 | Inquiry |





