ChemicalBook > Product Catalog >Analytical Chemistry >Standard >Pharmaceutical Impurity Reference Standards >Oxacillin Sodium Impurity G(EP)
Oxacillin Sodium Impurity G(EP)
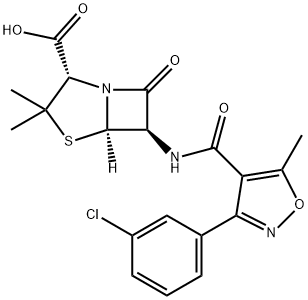
- CAS No.
- 724695-30-1
- Chemical Name:
- Oxacillin Sodium Impurity G(EP)
- Synonyms
- Oxacillin Impurity G;Oxacillin Impurity 21;Oxacillin EP Impurity G;Oxacillin Sodium Impurity G;Oxacillin sodium Impurity 12;Oxacillin Sodium Impurity G(EP);Benzylpenicillin EP Impurity G2;Histamine Dihydrochloride Impurity 3;Benzydamine Hydrochloride EP Impurity G2;(2S,5R,6R)-6-(3-(3-chlorophenyl)-5-methylisoxazole-4-carboxamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
- CBNumber:
- CB93370288
- Molecular Formula:
- C19H18ClN3O5S
- Molecular Weight:
- 435.88
- MOL File:
- 724695-30-1.mol
Oxacillin Sodium Impurity G(EP) Preparation Products And Raw materials
Raw materials
Preparation Products
Global( 26)Suppliers
| Supplier | Tel | Country | ProdList | Advantage | Inquiry |
|---|---|---|---|---|---|
| CLEARSYNTH LABS LTD. | +91-22-45045900 | Hyderabad, India | 6351 | 58 | Inquiry |
| SynZeal Research Pvt Ltd | +1 226-802-2078 | Gujarat, India | 6522 | 58 | Inquiry |
| China National Standard Pharmaceutical Corporation Limited | +8615391658522 | China | 11927 | 58 | Inquiry |
| Guangzhou PI PI Biotech Inc | +86-020-81716320 +86-13602409664 | China | 3635 | 55 | Inquiry |
| Hubei Qingbei Yunyan Pharmaceutical Technology Co., Ltd | 18162595016 | China | 9887 | 58 | Inquiry |
| Shanghai Kewel Chemical Co., Ltd. | 021-64609169 18901607656 | China | 9911 | 50 | Inquiry |
| Henan Vcommend Consultrading Co., Ltd. | 13393721940 | China | 10203 | 58 | Inquiry |
| Guangzhou Zhenlin Pharmaceutical Technology Co., Ltd | 18102736234 18102736234 | China | 7100 | 58 | Inquiry |
| Nantong Hanfang Biotechnology Co. , Ltd. | 18616537568 | China | 30968 | 58 | Inquiry |
| Nanjing wangzhixing Pharmaceutical Technology Co., Ltd | 15850510341 | China | 3620 | 58 | Inquiry |
724695-30-1(Oxacillin Sodium Impurity G(EP))Related Search:
Osimertinib Impurity J (AZD9291 Impurity J)
Osimertinib Impurity 3
Oxacillin Impurity H
Osimertinib Impurity 8
Oxacillin Sodium Impurity I(EP)
Osimertinib Dimer Impurity 2
Osimertinib Impurity I (AZD9291 Impurity I)
Oxacillin Sodium Impurity D(EP)
AZD-9291
3-[2,4-Bis((3S)-3-methylmorpholin-4-yl)pyrido[5,6-e]pyrimidin-7-yl]-N-methylbenzamide
chevron_left
1of4
chevron_right
Oxacillin Sodium Impurity G(EP)
Oxacillin Impurity G
Oxacillin EP Impurity G
(2S,5R,6R)-6-(3-(3-chlorophenyl)-5-methylisoxazole-4-carboxamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
(2S,5R,6R)-6-(3-(2-chlorophenyl)-5-methylisoxazole-4-carboxamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid compound with (2S,5R,6R)-6-(3-(3-chlorophenyl)-5-methylisoxazole-4-carboxamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid
Oxacillin sodium Impurity 12
Oxacillin Impurity 21
4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 6-[[[3-(3-chlorophenyl)-5-methyl-4-isoxazolyl]carbonyl]amino]-3,3-dimethyl-7-oxo-, (2S,5R,6R)-
Oxacillin Sodium Impurity G
Histamine Dihydrochloride Impurity 3
Benzydamine Hydrochloride EP Impurity G2
Benzylpenicillin EP Impurity G2
724695-30-1





