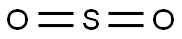Sulfur dioxide suppliers
Sulfur dioxide
- CAS:
- 7446-09-5
- MF:
- O2S
- MW:
- 64.06
Suppliers by country/region
Company Type
Properties
- Melting point:
- -73 °C (lit.)
- Boiling point:
- -10 °C (lit.)
- Density
- 1.25 g/mL at 25 °C (lit.)
- vapor density
- 2.26 (21 °C, vs air)
- vapor pressure
- 1779 mm Hg ( 21 °C)
- solubility
- soluble in H2O, EtOh, ethyl ether,chloroform
- form
- colorless gas
- color
- colorless
- Odor
- Pungent odor detectable at 0.3 to 5 ppm
- Odor Threshold
- 0.87ppm
- Water Solubility
- %H2O: 17.7 (0°C), 11.9 (15°C), 8.5 (25°C), 6.4 (35°C); % other solvents: 25, alcohol; 32, methanol [MER06]
- Merck
- 13,9061
- BRN
- 3535237
- Exposure limits
- TLV-TWA 2 ppm (~5 mg/m3) (ACGIH), 5 ppm (OSHA and MSHA); IDLH 100 ppm (NIOSH).
- Dielectric constant
- 17.6(-20℃)
- Stability:
- Stable. Non-corrosive when dry to common materials except zinc; corrosive when wet. Incompatible with strong reducing or oxidizing agents, moisture, zinc and its alloys.
Safety Information
- Symbol(GHS)



GHS04,GHS05,GHS06
- Signal word
- Danger
- Hazard statements
- H280-H314-H331
- Precautionary statements
- P260-P280-P303+P361+P353-P304+P340+P310-P305+P351+P338-P410+P403
- Hazard Codes
- T
- Risk Statements
- 23-34
- Safety Statements
- 9-26-36/37/39-45
- RIDADR
- UN 2037 2.3
- OEB
- B
- OEL
- TWA: 2 ppm (5 mg/m3), STEL: 5 ppm (13 mg/m3)
- WGK Germany
- 1
- RTECS
- WS4550000
- F
- 4.5-31
- DOT Classification
- 2.3, Hazard Zone C (Gas poisonous by inhalation)
- HazardClass
- 2.3
- Toxicity
- LC50 inhal (rat) 2520 ppm (6590 mg/m3; 1 h)
LCLO inhal (human) 1000 ppm (2600 mg/m3; 10 min)
PEL (OSHA) 5 ppm (13 mg/m3)
TLV-TWA (ACGIH) 2 ppm (5.2 mg/m3)
STEL (ACGIH) 5 ppm (13 mg/m3)
- IDLA
- 100 ppm
Use
Containers may explode in heat of fire or they may rupture and release irritating toxic Sulfur dioxide. Sulfur dioxide has explosive properties when Sulfur dioxide comes in contact with sodium hydride; potassium chlorate at elevated temperatures; ethanol; ether; zinc ethylsulfurinate at very cool temperatures (-15C); fluorine; chlorine trifluoride and chlorates. Sulfur dioxide will react with water or steam to produce toxic and corrosive fumes. When the liquid is heated Sulfur dioxide may release irritating, toxic Sulfur dioxide gas. Avoid ammonia, monocesium or monopotassium acetylide; dicesium monoxide; iron (II) oxide; tin oxide; lead (IV) oxide; chromium; manganese; molten sodium, powder aluminum and rubidium. Sulfur dioxide has explosive properties when Sulfur dioxide comes in contact with sodium hydride; potassium chlorate at elevated temperatures; ethanol; ether; zinc ethylsulfurinate at very cool temperatures (-15C); fluorine; chlorine trifluoride and chlorates. Sulfur dioxide will react with water or steam to produce toxic and corrosive fumes. Hazardous polymerization may not occur.
111 supplier list of "Sulfur dioxide"