Diamylamine suppliers
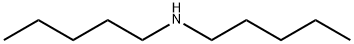
Diamylamine
- CAS:
- 2050-92-2
- MF:
- C10H23N
- MW:
- 157.3
Suppliers by country/region
Company Type
Properties
- Melting point:
- -44°C
- Boiling point:
- 202-203 °C(lit.)
- Density
- 0.767 g/mL at 25 °C
- vapor density
- 5.42 (vs air)
- vapor pressure
- 0.3 mm Hg ( 20 °C)
- refractive index
- n
20/D 1.4258(lit.)
- Flash point:
- 157 °F
- form
- clear liquid
- pka
- 11.25±0.19(Predicted)
- Specific Gravity
- 0.775 (25/4℃)
- color
- Colorless to Light yellow to Light orange
- Water Solubility
- Slightly soluble (0.1-1 g/100 mL)
- BRN
- 906746
- Dielectric constant
- 2.5
- Stability:
- Stable, but air-sensitive. Flammable. Incompatible with oxidizing agents.
Safety Information
- Symbol(GHS)




GHS09,GHS02,GHS05,GHS06
- Signal word
- Danger
- Hazard statements
- H226-H301+H311-H301+H311+H331-H314-H400
- Precautionary statements
- P261-P273-P280-P301+P310-P305+P351+P338-P310-P210-P233-P240-P241+P242+P243-P260-P264-P270-P301+P330+P331+P310-P303+P361+P353+P310+P363-P304+P340+P310-P305+P351+P338+P310-P370+P378-P403+P235-P405-P501
- Hazard Codes
- T,N,C
- Risk Statements
- 23/24/25-34-37/38-24-22-50/53
- Safety Statements
- 26-27-36/37/39-45-61-60
- RIDADR
- UN 2841 3/PG 3
- WGK Germany
- 3
- RTECS
- RZ9100000
- HS Code
- 2921.19.6190
- HazardClass
- 3.2
- PackingGroup
- III
Use
In contrast to n-amylamine, little information is available on diamylamine metabolism, particularly with respect to its suitability as a substrate for the amine oxidases. Generally, the rate of oxidation of secondary amines by monoamine oxidase is slower than that of primary amines (Beard and Noe 1981). In agreement, Yamada et al (1965) demonstrated that crystalline amine oxidase prepared from Aspergillus niger oxidized diamylamine very slowly with respect to n-amylamine. As with other secondary aliphatic amines, the propensity of diamylamine to form nitrosamines is of interest. It has been shown that treatment of diamylamine with nitrous acid in dilute aqueous solution gave optimum nitrosamine formation between pH 1 and 3, corresponding to stomach conditions (Sander et al 1968). When rats were fed a diet supplemented with sodium nitrite and secondary amines of low basicity, synthesis of nitrosamines in the stomach was observed. Malignant tumors arising through formation of nitrosamines in the stomach was demonstrated only when nitrite was present in the stomach concomitantly with secondary amines which readily formed carcinogenic nitrosamines.
81 supplier list of "Diamylamine"