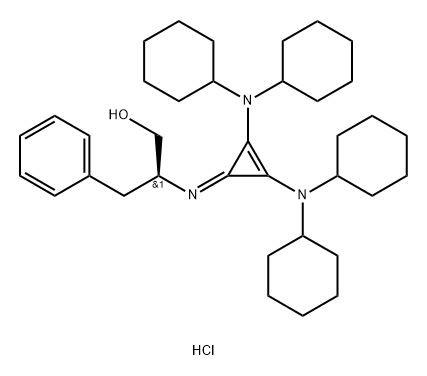
(S)-2-(2,3-Bis(dicyclohexylaMino)cyclopropeniMine)-3-phenylpropan-1-ol hydrochloride AldrichCPR
|
- $185 - $271
- Product name: (S)-2-(2,3-Bis(dicyclohexylaMino)cyclopropeniMine)-3-phenylpropan-1-ol hydrochloride AldrichCPR
- CAS: 1366421-67-1
- MF: C36H56ClN3O
- MW: 582.31
- EINECS:
- MDL Number:MFCD22417240
- Synonyms:(S)-2-(2,3-Bis(dicyclohexylaMino)cyclopropeniMine)-3-phenylpropan-1-ol hydrochloride AldrichCPR;(S)-2-(2,3-Bis(dicyclohexylamino)cyclopropenimine)-3-phenylpropan-1-ol hydrochloride;(βS)-β-[[2,3-bis(dicyclohexylamino)-2-cyclopropen-1-ylidene]amino]-benzenepropanol hydrochloride (1:1);Dicyclohexyl cyclopropenimine;Lambert Cyclopropenimine Catalyst;(S)-2-(2,3-Bis(dicyclohexylamino)cyclopropenimine)-3-phenylpropan-1-ol hydrochloride >=95%
2 prices
Selected condition:
Brand
- Sigma-Aldrich
Package
- 500mg
- 1g
- ManufacturerSigma-Aldrich
- Product number900811
- Product description(S)-2-(2,3-Bis(dicyclohexylamino)cyclopropenimine)-3-phenylpropan-1-ol hydrochloride ≥95%
- Packaging500mg
- Price$185
- Updated2024-03-01
- Buy
- ManufacturerSigma-Aldrich
- Product number900811
- Product description(S)-2-(2,3-Bis(dicyclohexylamino)cyclopropenimine)-3-phenylpropan-1-ol hydrochloride ≥95%
- Packaging1g
- Price$271
- Updated2024-03-01
- Buy
| Manufacturer | Product number | Product description | Packaging | Price | Updated | Buy |
|---|---|---|---|---|---|---|
| Sigma-Aldrich | 900811 | (S)-2-(2,3-Bis(dicyclohexylamino)cyclopropenimine)-3-phenylpropan-1-ol hydrochloride ≥95% | 500mg | $185 | 2024-03-01 | Buy |
| Sigma-Aldrich | 900811 | (S)-2-(2,3-Bis(dicyclohexylamino)cyclopropenimine)-3-phenylpropan-1-ol hydrochloride ≥95% | 1g | $271 | 2024-03-01 | Buy |
Properties
form :powder or solid
Safety Information
| Symbol(GHS): | ||||||||
|---|---|---|---|---|---|---|---|---|
| Signal word: | ||||||||
| Hazard statements: |
|
|||||||
| Precautionary statements: |
|
Description
Chiral cyclopropenimines are a highly effective new class of enantioselective Br?nsted base catalysts - the so-called “superbases” for enantioselective organocatalysis. Due to the prevalence of chemical reactions involving proton transfer as a key mechanistic event, Br?nsted bases have become indispensable tools for the practice of organic synthetic chemistry, capable of catalyzing proton transfer reactions enantioselectively for the production of optically enriched products. Catalyst is stored as co-salt for stability. Conversion of the HCl salt to free catalyst requires a simple wash with aqueous base. This is one of a suite of Br?nsted catalysts reported by Tristan Lambert and coworkers available through Sigma-Aldrich.Suppliers and manufacturers
Nantong HI-FUTURE Biology Co., Ltd.
Aladdin Scientific
Sigma-Aldrich
Energy Chemical
Nantong Hi-Future Biotechnology Co., Ltd
Shanghai Tachizaki Biomedical Research Center
Shanghai Yingxin laboratory equipment Co., Ltd
Shanghai Aladdin Biochemical Technology Co.,Ltd.
Jiangsu Aikon Biopharmaceutical R&D Co.,Ltd.





