| | 61116-33-4 Basic information More.. |
| Product Name: | asukamycin | | Synonyms: | asukamycin;7-Cyclohexyl-N-[5-hydroxy-5-[7-[(2-hydroxy-5-oxo-1-cyclopenten-1-yl)amino]-7-oxo-1,3,5-heptatrienyl]-2-oxo-7-oxabicyclo[4.1.0]hept-3-en-3-yl]-2,4,6-heptatrienamide;AM-1042;2,4,6-Heptatrienamide, 7-cyclohexyl-N-(5-hydroxy-5-(7-((2-hydroxy-5-oxo-1-cyclopenten-1-yl)amino)-7-oxo-1,3,5-heptatrienyl)-2-oxo-7-oxabicyclo(4.1.0)hept-3-en-3-yl)-;Brn 3639742;2,4,6-Heptatrienamide, 7-cyclohexyl-N-[(1S,5S,6R)-5-hydroxy-5-[(1E,3E,5E)-7-[(2-hydroxy-5-oxo-1-cyclopenten-1-yl)amino]-7-oxo-1,3,5-heptatrien-1-yl]-2-oxo-7-oxabicyclo[4.1.0]hept-3-en-3-yl]-, (2E,4E,6E)-;aviamycin | | CAS: | 61116-33-4 | | MF: | C31H34N2O7 | | MW: | 546.61 | | EINECS: | | | Mol File: | 61116-33-4.mol | 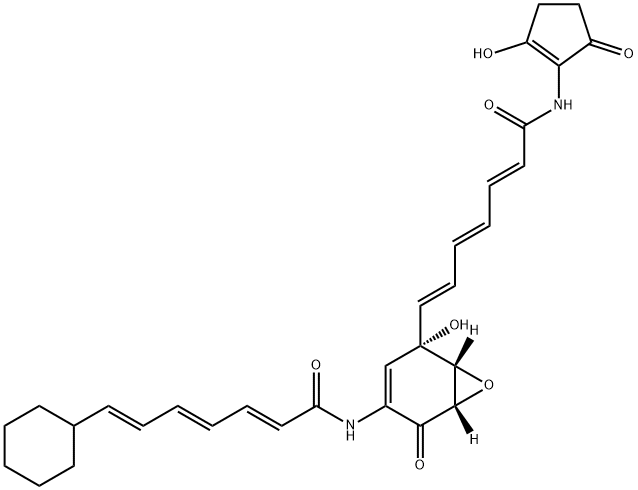 |
Use
Asukamycin is an unusual trienoic acid amide metabolite produced by Streptomyces nodosus, reported by Omura and colleagues at the Kitasato Institute, Japan, in 1976. Asukamycin belongs to the manumycin class that comprises two trienoic acid amides pivoted on a central cyclohexenone ring to give the molecule an unprecedented angular geometry. Asukamycin is active against Gram positive bacteria, tumor cell lines and protozoans, notably coccidia. Asukamycin’s cell toxicity is accompanied by activation of Caspases 3 and 8.
|
|
61116-33-4
Recommend Suppliers |
|