| | 849217-68-1 Basic information More.. |
| Product Name: | Cabozantinib | | Synonyms: | Cabozantinib(XL184Malate);Cabozantinib (XL184, BMS-907351);XL-186;1,1-cyclopropanedicarboxamide, n'-[4-[(6,7-dimethoxy-4-quinolinyl)oxy]phenyl]-n-(4-fluorophenyl)-;N-[4-[(6,7-Dimethoxy-4-quinolinyl)oxy]phenyl]-N'-(4-fluorophenyl)-1,1-cyclopropanedicarboxamide Cabozantinib (XL184, BMS-907351);Cabozantinib, >=98%;1-N-[4-(6,7-dimethoxyquinolin-4-yl)oxyphenyl]-1-N'-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide;N-(4-(6,7-dimethoxyquinolin-4-yloxy)phenyl)-N-(4-fluorophenyl)cyclopropane-1,1-dicarboxamide Cabozantinib | | CAS: | 849217-68-1 | | MF: | C28H24FN3O5 | | MW: | 501.51 | | EINECS: | 692-846-8 | | Mol File: | 849217-68-1.mol | 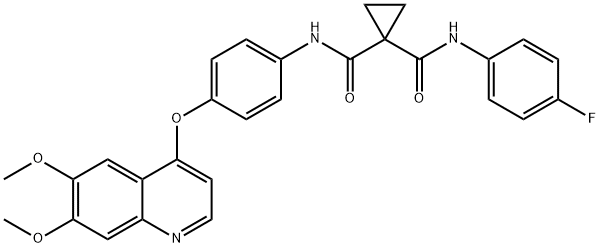 |
Use
In clinical trials, common adverse effects caused by cabozantinib include diarrhea, stomatitis, palmar-plantarrythrodysesthesia syndrome (PPES), and loss of weight, loss of appetite, nausea, fatigue, oral pain, taste disorders, high blood pressure, abdominal pain and constipation.
The most common laboratory abnormalities ( 25%) were increased aspartate aminotransferase, alanine aminotransferase, lymphopenia, increased alkaline phosphatase level, hypocalcemia, neutropenia, thrombocytopenia, hypophosphatemia and hyperbilirubinemia.
For patients receiving cabozantinib, 57% of patients have gotten increased level of thyroid stimulating hormone after initial dose, compared with patients receiving placebo, increased by 19%.
Information regarding to the anticancer drug, mechanism of action, pharmacokinetics, side effects, and drug interactions of cabozantinib were compiled and edited by Yan Shi from Chemicalbook. (2015-10-23)
- Cabozantinib
-

- US $0.00 / g
- 2024-08-27
- CAS:849217-68-1
- Min. Order: 1g
- Purity: 99%min
- Supply Ability: 1000g
|
|
849217-68-1
Recommend Suppliers |
|