| | 79277-27-3 Basic information More.. |
| Product Name: | Thifensulfuron methyl | | Synonyms: | PINNACLE (TM);2-thiophenecarboxylicacid,3-(((((4-methoxy-6-methyl-1,3,5-triazin-2-yl)amino);dpx-m6316;methyl3-(4-methoxy-6-methyl-1,3,5-triazin-2-ylcarbamoylsulfamoyl)-2-thenoate;thiameturon-methyl;METHYL 3-[[[[(4-METHOXY-6-METHYL-1,3,5-TRIAZIN-2-YL)AMINO]CARBONYL]AMINO]-SULFONYL]-2-THIOPHENECARBOXYLATE;METHYL 3-(4-METHOXY-6-METHYL-1,3,5-TRIAZIN-2-YL-CARBAMOYLSULFAMOYL)-THIOPHEN-2-CARBOXYLATE;DPX-M6316(Du Pont) | | CAS: | 79277-27-3 | | MF: | C12H13N5O6S2 | | MW: | 387.39 | | EINECS: | 616-673-4 | | Mol File: | 79277-27-3.mol | 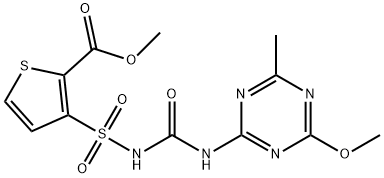 |
Use
The hydrolytic degradation of thifensulfuron methyl is
pH dependent and, in alkaline condition, specifically
yields the corresponding free acid. Primary
degradation cleaves the sulfonylurea moiety to give
two typical hydrolyzed products, sulfonamide and
aminotriazine analogs, derived from thifensulfuron
methyl in acidic and neutral conditions. Hydrolysis of
the ester linkage proceeds in mammals, plants, soils, and also chemical hydrolysis, and opening of the
triazine ring occurs to yield the acetyltriuret analog
identified. On the other hand, by hydrolysis, O-
demethylated thifensulfuron methyl undergoes opening
of the triazine ring to give the corresponding
acetyltriuret analog. Under photolytic conditions,
methyl 3-(4-methoxy-6-methyl-1,3,5-triazin-2-
yl)aminothiophene-2-carboxylate is identified.
- Thifensulfuron methyl
-

- US $0.00 / kg
- 2024-08-27
- CAS:79277-27-3
- Min. Order: 100kg
- Purity: 75%
- Supply Ability: 20tons
- Thifensulfuron methyl
-

- US $0.00 / KG
- 2023-09-06
- CAS:79277-27-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 500000kg
- Thifensulfuron methyl
-

- US $0.00 / KG
- 2023-01-31
- CAS:79277-27-3
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 50000kg/week
|
|
79277-27-3
Recommend Suppliers |
|