| | 2050-92-2 Basic information More.. |
| Product Name: | Diamylamine | | Synonyms: | 1-Pentanamine,N-pentyl-;amine,dipentyl;diamylamine(melanged’isomeres);diamylamine(mixedisomers);DIAMYLAMINE, TECH;N,N-Diamylamine;Dipentylamine,99%;DipentylaMine, Mixture of isoMers 99% | | CAS: | 2050-92-2 | | MF: | C10H23N | | MW: | 157.3 | | EINECS: | 218-108-3 | | Mol File: | 2050-92-2.mol | 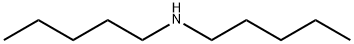 |
Use
In contrast to n-amylamine, little information is available on diamylamine metabolism,
particularly with respect to its suitability as a substrate for the amine
oxidases. Generally, the rate of oxidation of secondary amines by monoamine
oxidase is slower than that of primary amines (Beard and Noe 1981). In agreement,
Yamada et al (1965) demonstrated that crystalline amine oxidase prepared
from Aspergillus niger oxidized diamylamine very slowly with respect to n-amylamine.
As with other secondary aliphatic amines, the propensity of diamylamine to
form nitrosamines is of interest. It has been shown that treatment of diamylamine
with nitrous acid in dilute aqueous solution gave optimum nitrosamine formation
between pH 1 and 3, corresponding to stomach conditions (Sander et al 1968).
When rats were fed a diet supplemented with sodium nitrite and secondary amines
of low basicity, synthesis of nitrosamines in the stomach was observed. Malignant
tumors arising through formation of nitrosamines in the stomach was demonstrated
only when nitrite was present in the stomach concomitantly with secondary
amines which readily formed carcinogenic nitrosamines.
|
|
2050-92-2
Recommend Suppliers |
|