|
|
| | (20S)-20-Hydroxyvoaluteine Basic information |
| Product Name: | (20S)-20-Hydroxyvoaluteine | | Synonyms: | (20S)-20-Hydroxyvoaluteine;Montanine;Montanine【Tabernaemontana】;Voacristine pseudoindoxyl;Spiro[2H-1,6-methanoquinoline-4(4aH),2'-[2H]indole]-4a-carboxylic acid, 1',3,3',5,6,7,8,8a-octahydro-8-[(1S)-1-hydroxyethyl]-5'-methoxy-3'-oxo-, methyl ester, (1R,2'R,4aR,6S,8R,8aR)- | | CAS: | 18646-15-6 | | MF: | C22H28N2O5 | | MW: | 400.47 | | EINECS: | | | Product Categories: | | | Mol File: | 18646-15-6.mol | 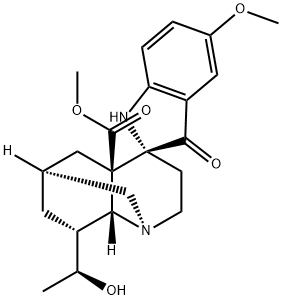 |
| | (20S)-20-Hydroxyvoaluteine Chemical Properties |
| Melting point | l37-8°C | | Boiling point | 564.6±50.0 °C(Predicted) | | density | 1.35±0.1 g/cm3(Predicted) | | pka | 14.93±0.20(Predicted) |
| | (20S)-20-Hydroxyvoaluteine Usage And Synthesis |
| Description | This alkaloid occurs in the leaf extract from Tabernaernontana rupicoia Benth. It
is an amorphous substance which cannot be crystallized. The ultraviolet spectrum
has absorption maxima at 228 and 410 mil with a shoulder at 260 mil. It yields
crystalline salts with mineral acids, e.g. the hydrochloride, m.p. 258-9°C (dec.)
and the hydrobromide, m.p. 273-4°C (dec.); [α]26D - 206° (c 1.21, H20). The
structure has been established from the infrared and NMR spectra. | | References | Niemann, Kessel.,!. Org. Chern., 31,2265 (1966) |
| | (20S)-20-Hydroxyvoaluteine Preparation Products And Raw materials |
|