| Company Name: |
Shanghai Hongye Biotechnology Co. Ltd
|
| Tel: |
400-9205774 |
| Email: |
sales@glpbio.cn |
| Products Intro: |
Product Name:JWH 200 2’-naphthyl isomer
CAS:133438-66-1
Purity:>98% Package:1mg;10mg;50mg;100mg;
|
| Company Name: |
ChemeGen(Shanghai) Biotechnology Co.,Ltd.
|
| Tel: |
18818260767 |
| Email: |
sales@chemegen.com |
| Products Intro: |
Product Name:JWH 200 2'-naphthyl isomer
CAS:133438-66-1
Purity:98% Package:10 mg;50 mg;100 mg;500 mg;1 g;5 g;10 g
|
| Company Name: |
Cayman Chemical Company
|
| Tel: |
(800) 364-9897 |
| Email: |
cayman@caymanchem.com |
| Products Intro: |
Product Name:JWH 200 2'-naphthyl isomer
CAS:133438-66-1
Package:>=98% Remarks:9000897
|
| Company Name: |
Resenbio
|
| Tel: |
029-86354885 |
| Email: |
sales@xarxbio.com |
| Products Intro: |
Product Name:JWH 200 2'-naphthyl isomer
CAS:133438-66-1
|
|
| | JWH 200 2'-naphthyl isomer Basic information |
| Product Name: | JWH 200 2'-naphthyl isomer | | Synonyms: | JWH 200 2'-naphthyl isomer;[1-(2-morpholin-4-ylethyl)indol-3-yl]-naphthalen-2-ylmethanone;JWH 200 2’Methanone, [1-[2-(4-morpholinyl)ethyl]-1H-indol-3-yl]-2-naphthalenyl- | | CAS: | 133438-66-1 | | MF: | C25H24N2O2 | | MW: | 384.47 | | EINECS: | | | Product Categories: | | | Mol File: | 133438-66-1.mol | 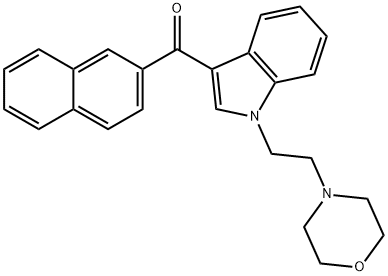 |
| | JWH 200 2'-naphthyl isomer Chemical Properties |
| solubility | DMF: 0.30 mg/ml; DMSO: 0.15 mg/ml | | form | A crystalline solid |
| | JWH 200 2'-naphthyl isomer Usage And Synthesis |
| Description | JWH 200 is a 3-indolyl-1-naphthylmethane that acts as a cannabinoid (CB) receptor agonist, binding to the CB1 receptor with high affinity (IC50 = 42 nM).1 This synthetic cannabinoid has been identified as an adulterant of herbal blends.2 JWH 200 2’-naphthyl isomer differs structurally from JWH 200 by having the naphthyl group attached at the 2’ position. The biological and toxicological properties of this compound have not been characterized. This product is intended for forensic purposes. | | References | 1. Huffman, J.W., Mabon, R., Wu, M.J., et al. 3-indolyl-1-naphthylmethanes: New cannabimimetic indoles provide evidence for aromatic stacking interactions with the CB1 cannabinoid receptor Bioorgan. Med. Chem. 11(4),539-549(2003).
2. Kikura-Hanajiri, R., Uchiyama, N., and Goda, Y. Survey of current trends in the abuse of psychotropic substances and plants in Japan Leg. Med. (Tokyo) 13(3),109-115(2011). |
| | JWH 200 2'-naphthyl isomer Preparation Products And Raw materials |
|