|
|
| | Hexachloroparaldehyde Basic information |
| Product Name: | Hexachloroparaldehyde | | Synonyms: | 2,4,6-tris(dichloromethyl)-1,3,5-trioxane;Einecs 241-375-2;1,3,5-Trioxane, 2,4,6-tris(dichloromethyl)- | | CAS: | 17352-16-8 | | MF: | C6H6Cl6O3 | | MW: | 338.83 | | EINECS: | 241-375-2 | | Product Categories: | | | Mol File: | 17352-16-8.mol | 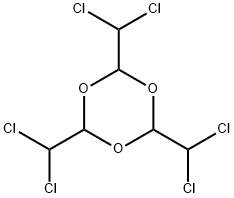 |
| | Hexachloroparaldehyde Chemical Properties |
| Boiling point | 376.9±37.0 °C(Predicted) | | density | 1.651±0.06 g/cm3(Predicted) |
| | Hexachloroparaldehyde Usage And Synthesis |
| Description | Hexachloroparaldehyde [17352-16-8], 2,4,6-
tris(dichloromethyl)-1,3,5-trioxane, C6H6Cl6O3,
Mr338.83, mp 131–132℃, colorless crystals, bp
210–220℃ with the formation of small amounts of
dichloroacetaldehyde. | | Preparation | Hexachloroparaldehyde is made by treating
dichloroacetaldehyde with such Lewis acids as
antimony trichloride, iron(III) chloride, aluminum trichloride,tin(IV) chloride, or boron trifluoride. Hexachloroparaldehyde is also
formed by the direct chlorination of paraldehyde with 6–7 mol of chlorine at 35℃ under anhydrous conditions. It forms dichloroacetaldehyde
hydrate in the presence of aqueous acids |
| | Hexachloroparaldehyde Preparation Products And Raw materials |
|