|
|
| | spirilloxanthin Basic information |
| Product Name: | spirilloxanthin | | Synonyms: | spirilloxanthin;all-trans-3,3',4,4'-Tetradehydro-1,1',2,2'-tetrahydro-1,1'-dimethoxylycopene;all-trans-Rhodoviolascin;all-trans-Spirilloxanthin;Rhodoviolascin;Rodoviolascin;ψ,ψ-Carotene, 3,3',4,4'-tetradehydro-1,1',2,2'-tetrahydro-1,1'-dimethoxy- | | CAS: | 34255-08-8 | | MF: | C42H60O2 | | MW: | 596.92 | | EINECS: | | | Product Categories: | | | Mol File: | 34255-08-8.mol | 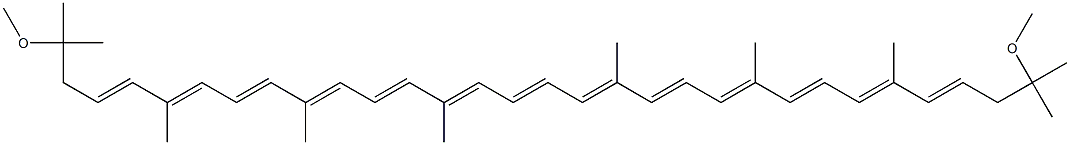 |
| | spirilloxanthin Chemical Properties |
| Melting point | 218° | | Boiling point | 701.1±53.0 °C(Predicted) | | density | 0.917±0.06 g/cm3(Predicted) |
| | spirilloxanthin Usage And Synthesis |
| Definition | ChEBI: A carotenoid ether that is (3E,3'E)-1,1'-dihydroxy-3,3',4,4'-tetradehydro-1,1',2,2'-tetrahydro-psi,psi-carotene in which both hydroxyl hydrogens are replaced by methyl groups. | | Purification Methods | Spirilloxanthin crystallises from CHCl3/pet ether, acetone/pet ether, *C6H6/pet ether or *C6H6 as deep red spindle-like crystals. Purify it also by chromatography on a column of CaCO3/Ca(OH)2 mixture, Ca(OH)2 or deactivated alumina and elute it with a *C6H6/MeOH mixture. It gives a blue colour with SbCl3 in CHCl3 with at 642nm. [Polgar et al. Arch max Biochem Biophys 5 243 1944, Synthesis: Surmatis & Ofner J Org Chem 28 2735 1963, Karrer & Koenig Helv Chim Acta 23 460 1940.] Store it in the dark in an inert atmosphere at -20o. [Beilstein 1 III 2297, 1 IV 2750.] |
| | spirilloxanthin Preparation Products And Raw materials |
|