| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
010-82848833 400-666-7788 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95%
CAS:364795-64-2
Purity:min. 95% Package:10mg;50mg
|
| Company Name: |
Alfa Chemistry
|
| Tel: |
1-516-6625404 |
| Email: |
support@alfa-chemistry.com |
| Products Intro: |
Product Name:Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95%
CAS:364795-64-2
Package:1g;10g;100g;1KG;5KG
|
| Company Name: |
Tianjin Kailiqi Biotechnology Co., Ltd.
|
| Tel: |
15076683720 |
| Email: |
klq@cw-bio.com |
| Products Intro: |
Product Name:Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II)
CAS:364795-64-2
Purity:大于98% Package:1g,5g,10g,25g根据客户需要分装 Remarks:Not For Human Use, Lab Use Only.
|
|
| | Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95% Basic information | | Reaction |
| Product Name: | Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95% | | Synonyms: | Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95% | | CAS: | 364795-64-2 | | MF: | C62H66Cl2N2P2Ru | | MW: | 1073.14 | | EINECS: | | | Product Categories: | | | Mol File: | 364795-64-2.mol | ![Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95% Structure](CAS/20180808/GIF/364795-64-2.gif) |
| | Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95% Chemical Properties |
| form | Powder | | color | cream colored | | Sensitive | air sensitive |
| | Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95% Usage And Synthesis |
| Reaction |
- The Noyori [(diphosphine) RuCl2 (diamine)] catalysts containing the chiral ligand Xylyl-Phanephos display exceptional activity and enantioselectivity in the asymmetric hydrogenation of a wide range of aromatic, heteroaromatic and α,β-unsaturated ketones.
- Reactions are performed under mild conditions at room temperature and typically at low H2 pressures of 2-10 bar. High substrate concentrations of up to 40% w/v are tolerated.
- Molar substrate/catalyst ratios of up to 100,000/1 are achieved with excellent reactivity and enantioselectivity using commercial grade substrates and solvents.
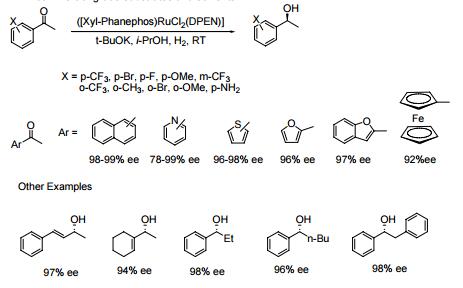
|
| | Dichloro[(S)-(+)-4,12-bis(di(3,5-xylyl)phosphino)-[2.2]-paracyclophane][(1R,2R)-(+)-1,2-diphenylethylenediamine]ruthenium(II), min. 95% Preparation Products And Raw materials |
|