|
|
| | Epi-N-Quinyl-N’-bis(3,5-trifluoromethyl)
phenylthiourea Basic information |
| Product Name: | Epi-N-Quinyl-N’-bis(3,5-trifluoromethyl)
phenylthiourea | | Synonyms: | N-[3,5-Bis(trifluoromethyl)phenyl]-N'-[(8a,9S)-6'-methoxy-9-cinchonanyl]thiourea;N-[3,5-Bis(trifluoromethyl)phenyl]-N'-[(8a,9S)-6'-methoxy-9-cinchonanyl]thiourea 90%; 1-[3,5-BIS(TRIFLUOROMETHYL)PHENYL]-3-[(S)(6-METHOXY-4-QUINOLINYL)-[(2S,4S,5R)-5-VINYL-1-AZA-BICYCLO[2.2.2]OCT-2-YL]METHYL]THIOUREA;1-[3,5-bis(trifluoromethyl)phenyl]-3-[(S)-[(1S,2S,4S,5R)-5-ethenyl-1-azabicyclo[2.2.2]octan-2-yl](6-methoxyquinolin-4-yl)methyl]thiourea;N-[3,5-Bis(trifluoromethyl)phenyl]-N'-[(8α,9S)-6'-methox
ycinchonan-9-yl]thiourea,99%d.e.;1-(3,5-Bis(trifluoromethyl)phenyl)-3-((6-methoxyquinolin-4-yl)((1S,2R,4S,5R)-5-vinylquinuclidin-2-yl)methyl)thiourea;Epi-N-Quinyl-N’-bis(3,5-trifluoromethyl)
phenylthiourea;N-[3,5-bis(trifluoroMethyl)phenyl]-N'-[(8α,9S)-6'-Methoxycinchonan-9-yl]- Thiourea | | CAS: | 852913-16-7 | | MF: | C29H28F6N4OS | | MW: | 594.61 | | EINECS: | | | Product Categories: | | | Mol File: | 852913-16-7.mol | 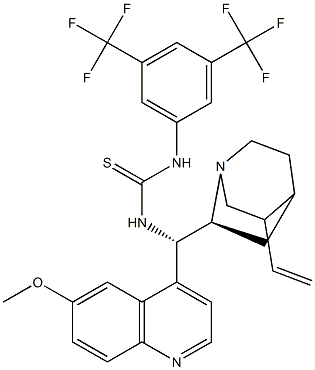 |
| | Epi-N-Quinyl-N’-bis(3,5-trifluoromethyl)
phenylthiourea Chemical Properties |
| Boiling point | 598.2±60.0 °C(Predicted) | | density | 1.39±0.1 g/cm3(Predicted) | | pka | 11.39±0.70(Predicted) |
| Hazard Codes | T | | Risk Statements | 25 | | Safety Statements | 45 | | RIDADR | UN 2811 6.1 / PGIII | | HS Code | 29339900 |
| | Epi-N-Quinyl-N’-bis(3,5-trifluoromethyl)
phenylthiourea Usage And Synthesis |
| Chemical Properties | Solid | | Uses | N-[3,5-Bis(trifluoromethyl)phenyl]-N′-[(8a,9S)-6′-methoxy-9-cinchonanyl]thiourea is a bifunctional cinchona organocatalyst, which can be used to synthesize:
- Stereoselective diaryl(nitro)butanone via enantioselective Michael addition of nitromethane to chalcones.
- Enantioselective β-amino acids via asymmetric Mannich reaction of malonates with aryl and alkyl imines.
- The synthesis of 3-indolylmethanamines by the reaction of indoles with imines via asymmetric Friedel-Crafts reaction.
- The enantioselective conjugate addition of active methylene compounds to enones to obtain the corresponding addition products.
|
| | Epi-N-Quinyl-N’-bis(3,5-trifluoromethyl)
phenylthiourea Preparation Products And Raw materials |
|