- Iron hydroxide oxide
-

- $50.00 / 1KG
-
2021-08-31
- CAS:20344-49-4
- Min. Order: 1Kg/Bag
- Purity: 99.99%
- Supply Ability: 20 tons/month
- Iron hydroxide oxide
-

- $1.00 / 1KG
-
2020-01-09
- CAS:83-42-1
- Min. Order: 1KG
- Purity: 85.0-99.8%
- Supply Ability: 20tons
- 2-Chloro-6-nitrotoluene
-

- $1.00 / 1KG
-
2020-01-09
- CAS:20344-49-4
- Min. Order: 1KG
- Purity: 85.0-99.8%
- Supply Ability: 20tons
|
| | Iron hydroxide oxide Basic information |
| | Iron hydroxide oxide Chemical Properties |
| | Iron hydroxide oxide Usage And Synthesis |
| Chemical Properties | reddish-brown crystalline solid | | Uses | Iron(III) hydroxide, γ-phase is used in purifying water and as an absorbent in chemical processes. It is involved in the preparation of iron oxide-hydroxides nanoparticles, which is used as a very good adsorbent for lead removal from aquatic media. It is also used as a pigment, as a catalyst and in aquarium water treatment as a phosphate binder. Further, it is employed in the paints-lacquers-varnishes industry. | | Uses | In purifying water; as absorbent in chemical processing; as pigment; as catalyst. | | Preparation |
A cold solution of 810 g of Fe(NO3)3?9H2O in two liters of water is poured slowly, with vigorous stirring, into an ammonia solution prepared by dissolving 120 g of gaseous NH3 in two liters of water (cooling necessary). The hydroxide which precipitates is amorphous to x-ray analysis. It is washed by stirring at least five times with eight-liter portions of cold water, each portion being decanted as completely as possible. The residual slurry is then stirred with sufficient cone. KOH solution to give a mixture approximately 2N and allowed to stand for 3-4 hours. Finally 100℃ steam is bubbled through for two hours. The precipitate is thereby transformed completely into bright-yellow α-FeO(OH), which shows a crystalline x-ray diffraction pattern.
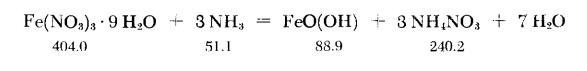
| | General Description | contains varying amounts of MgO2, SiO2, CaO, Al2O3 |
| | Iron hydroxide oxide Preparation Products And Raw materials |
|