| Company Name: |
Shanghai EFE Biological Technology Co., Ltd.
|
| Tel: |
021-65675885 18964387627 |
| Email: |
info@efebio.com |
| Products Intro: |
Product Name:17-phenyl trinor Prostaglandin F2α-d4
CAS:58976-50-4
Purity:95% Package:25μg;50μg;100μg
|
| Company Name: |
ChemeGen(Shanghai) Biotechnology Co.,Ltd.
|
| Tel: |
18818260767 |
| Email: |
sales@chemegen.com |
| Products Intro: |
Product Name:17-phenyl trinor Prostaglandin F2α-d4
CAS:58976-50-4
Purity:98% Package:10 mg;50 mg;100 mg;500 mg;1 g;5 g;10 g
|
|
| | 17-phenyl trinor Prostaglandin F2α-d4 Basic information |
| Product Name: | 17-phenyl trinor Prostaglandin F2α-d4 | | Synonyms: | 17-phenyl trinor Prostaglandin F2α-d4;YFHHIZGZVLHBQZ-QOHQNJFUSA-N;Bimatoprost Acid-d4;17-phenyl trinor Prostaglandin F2α-d4;Bimatoprost Impurity 5-d4;Bimatoprost, free acid-d4 | | CAS: | 58976-50-4 | | MF: | C23H32O5 | | MW: | 388.5 | | EINECS: | | | Product Categories: | | | Mol File: | 58976-50-4.mol | 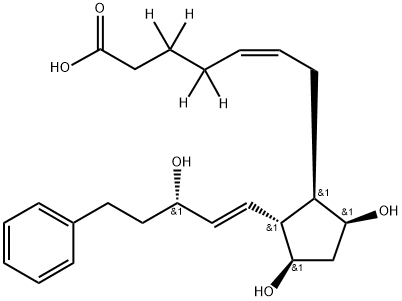 |
| | 17-phenyl trinor Prostaglandin F2α-d4 Chemical Properties |
| storage temp. | Store at -20°C | | solubility | DMF: 25 mg/ml,DMSO: 25 mg/ml,Ethanol: 30 mg/ml,PBS (pH 7.2): .5 mg/ml | | form | Oil | | color | White to off-white |
| | 17-phenyl trinor Prostaglandin F2α-d4 Usage And Synthesis |
| Definition | ChEBI: 17-phenyl trinor Prostaglandin F2alpha-d4 is a prostanoid. | | Biological Activity | 17-phenyl trinor Prostaglandin F2α-d4 (17-phenyl trinor PGF2α-d4) contains four deuterium atoms at the 3, 3', 4, and 4' positions. It is intended for use as an internal standard for the quantification of 17-phenyl trinor PGF2α by GC- or LC-mass spectrometry. 17-phenyl trinor PGF2α is a metabolically stable analog of PGF2α and is a potent agonist for the FP receptor. It binds to the FP receptor on ovine luteal cells with a relative potency of 756% compared to that of PGF2α.1 At the rat recombinant FP receptor expressed in CHO cells 17-phenyl trinor PGF2α inhibits PGF2α binding with a Ki of 1.1 nM.2 The isopropyl ester of 17-phenyl trinor PGF2α-d4 is slightly better than PGF2α isopropyl ester in reducing the intraocular pressure in the cat eye without any irritation.3 | | References | 1.Balapure, A.K., Rexroad, C.E., Jr., Kawada, K., et al.Structural requirements for prostaglandin analog interaction with the ovine corpus luteum prostaglandin F2α receptorBiochem. Pharmacol.38(14)2375-2381(1989)
2.Lake, S., Gullberg, H., Wahlqvist, J., et al.Cloning of the rat and human prostaglandin F2α receptors and the expression of the rat prostaglandin F2α receptorFEBS Letters355317-325(1994)
3.Stjernschantz, J., and Resul, B.Phenyl substituted prostaglandin analogs for glaucoma treatmentDrugs of the Future17691-704(1992) |
| | 17-phenyl trinor Prostaglandin F2α-d4 Preparation Products And Raw materials |
|