Rimegepant manufacturers
- BMS-927711
-

- $0.00 / 1g
-
2024-03-12
- CAS:1289023-67-1
- Min. Order: 1g
- Purity: 98% HPLC
- Supply Ability: 100kg
- Rimegepant
-

- $20.00 / 1KG
-
2023-09-05
- CAS:1289023-67-1
- Min. Order: 1KG
- Purity: 0.99
- Supply Ability: 20
- Rimegepant
-
- $0.00 / 1KG
-
2023-07-15
- CAS:1289023-67-1
- Min. Order: 0.1KG
- Purity: 98%
- Supply Ability: 1000KGS
|
| | Rimegepant Basic information |
| Product Name: | Rimegepant | | Synonyms: | 4-(2,3-Dihydro-2-oxo-1H-imidazo[4,5-b]pyridin-1-yl)-1-piperidinecarboxylic acid (5S,6S,9R)-5-amino-6-(2,3-difluorophenyl)-6,7,8,9-tetrahydro-5H-cyclohepta[b]pyridin-9-yl ester;Rimegepant;CS-2806;BMS-927711(Rimegepant);BMS 92771;RIMEGEPANT;BMS-927711;BMS927711;BMS 92771;(5S,6S,9R)-5-amino-6-(2,3-difluorophenyl)-6,7,8,9-tetrahydro-5H-cyclohepta[b]pyridin-9-yl 4-(2-oxo-2,3-dihydro-1H-imidazo[4,5-b]pyridin-1-yl)piperidine-1-carboxylate;1-Piperidinecarboxylic acid, 4-(2,3-dihydro-2-oxo-1H-imidazo[4,5-b]pyridin-1-yl)-, (5S,6S,9R)-5-amino-6-(2,3-difluorophenyl)-6,7,8,9-tetrahydro-5H-cyclohepta[b]pyridin-9-yl ester | | CAS: | 1289023-67-1 | | MF: | C28H28F2N6O3 | | MW: | 534.56 | | EINECS: | | | Product Categories: | API;1289023-67-1 | | Mol File: | 1289023-67-1.mol | 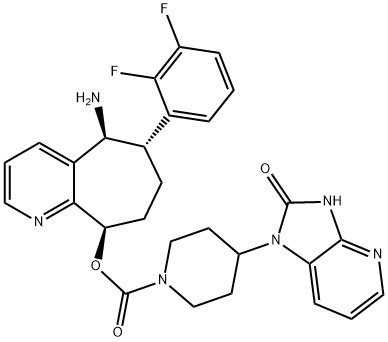 |
| | Rimegepant Chemical Properties |
| density | 1.45±0.1 g/cm3(Predicted) | | storage temp. | Store at -20°C | | solubility | Soluble in DMSO | | form | A crystalline solid | | pka | 12.03±0.20(Predicted) |
| | Rimegepant Usage And Synthesis |
| Description | Rimegepant (Nurtec ODT)(FDA approved in February 2020) is an ODT available in a single strength of 75 mg for the acute treatment of migraine with or without aura in adults.No more than 75 mg should be taken in a 24-hour period. while there are no contraindications,concomitant administration with CYP3A4 inhibitors should be avoided.The most common adverse reaction is nausea, occurring in few patients (rimegepant PI, 2020). In clinical trials, a single 75-mg dose of rimegepant demonstrated superiority over placebo with regard to freedom from pain (21% in the rimegepant group vs.11% in the placebo group) and freedom from the most bothersome symptom (35% vs. 27%) at 2 hours post dose (Croop et al., 2019). Rimegepant is not approved for migraine prevention, although it is presently in late-stage clinical trials assessing it for that indication(NCT03732638,2020). | | Uses | Rimegepant is an oral calcitonin gene-related peptide (CGRP) receptor antagonist used for the treatment of migraine. |
| | Rimegepant Preparation Products And Raw materials |
| Raw materials | 1-Piperidinecarboxylic acid, 4-(2,3-dihydro-2-oxo-1H-imidazo[4,5-b]pyridin-1-yl)-, (5S,6S,9R)-5-azido-6-(2,3-difluorophenyl)-6,7,8,9-tetrahydro-5H-cyclohepta[b]pyridin-9-yl ester |
|