| Company Name: |
J & K SCIENTIFIC LTD.
|
| Tel: |
010-82848833 400-666-7788 |
| Email: |
jkinfo@jkchemical.com |
| Products Intro: |
Product Name:(8α;, 9S)-(+)-9-Amino-cinchonan-6'-ol, min. 90%
CAS:960050-59-3
Purity:min. 90% Package:250mg;50mg
|
| Company Name: |
Daicel Chiral Technologies (China)CO.,LTD
|
| Tel: |
021-50460086-9 15921403865 |
| Email: |
han_yajun@dctc.daicel.com |
| Products Intro: |
Product Name:(8α,9S)-9-Aminocinchonan-6'-ol
CAS:960050-59-3
Purity:95%/98% Package:100MG;100G; 1KG Remarks:135374
|
| Company Name: |
Shanghai Chiral bio-compound co., Ltd.
|
| Tel: |
021-5068 3667/17749785980/1029026415; 17749785980 |
| Email: |
1029026415@qq.com |
| Products Intro: |
Product Name:(8α,9S)-9-amino-Cinchonan-6'-ol
CAS:960050-59-3
Purity:97% Package:100mg,1g,100g,1kg
|
| Company Name: |
LaaJoo
|
| Tel: |
021-60702684 18516024827 |
| Email: |
huang.jiayi@sinocompound.com |
| Products Intro: |
CAS:960050-59-3
Purity:98%,99%e.e. Package:50mg;100mg
|
|
| | (8α,9S)-9-aMino-Cinchonan-6'-ol Basic information | | Reaction |
| Product Name: | (8α,9S)-9-aMino-Cinchonan-6'-ol | | Synonyms: | (8α,9S)-9-aMino-Cinchonan-6'-ol;(8α, 9S)-(+)-9-AMino-cinchonan-6'-ol, Min. 90%;(8Α,9S)-(+)-9-AMINO-CINCHONAN-6'-OL,MIN.90%;9S)-(+)-9-Amino-cinchonan-6'-ol, min. 90%;9-amino-(9-deoxy)epi-6-hydroxycinchonidine;Cinchonan-6'-ol, 9-amino-, (8α,9S)-;4-((S)-Amino((1S,2S,4S,5R)-5-vinylquinuclidin-2-yl)methyl)quinolin-6-ol;(8α,9S)-9-amino-Cinchonan-6'-ol | | CAS: | 960050-59-3 | | MF: | C19H23N3O | | MW: | 309.41 | | EINECS: | | | Product Categories: | | | Mol File: | 960050-59-3.mol | 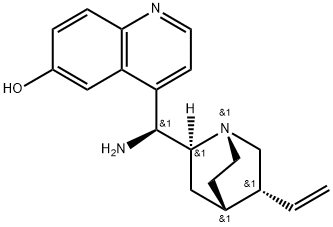 |
| | (8α,9S)-9-aMino-Cinchonan-6'-ol Chemical Properties |
| alpha | +107.9° (c 1.0, CHCl3) | | Boiling point | 501.7±45.0 °C(Predicted) | | density | 1.24±0.1 g/cm3(Predicted) | | pka | 8.92±0.40(Predicted) | | form | Powder | | color | off-white to pale brown | | Stability: | store cold |
| | (8α,9S)-9-aMino-Cinchonan-6'-ol Usage And Synthesis |
| Reaction | 1. [2+3]-dipolar cycloaddition of cyclic enones.
2. Activation of α,β-unsaturated carbonyl compounds:
a) vinylogous α-ketol rearrangement.
b) vinylogous Michael addition of β-substituted α,β-unsaturated cyclohexanones.
3. Michael addition
a) α-nitroacetate to α,β-unsaturated ketones.
b) Diastereodivergent Michael addition to α-substituted, α,β-unsaturated ketones.
4. α-benzoyloxylation of α-branched aldehydes.
5. [4+2]-cycloadditions of β-substituted α,β-unsaturated cyclohexanones with polyconjugated malonitriles.
6. Vinylogous organocascade catalysis with control of remote stereochemistry in the synthesis of spirocyclic oxindoles.
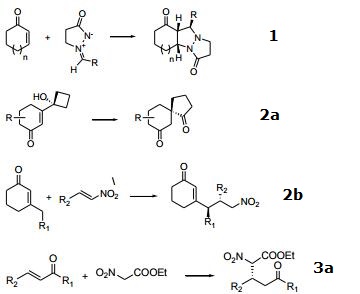
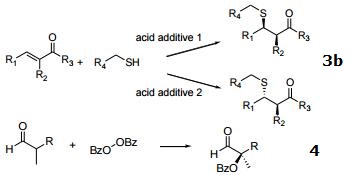
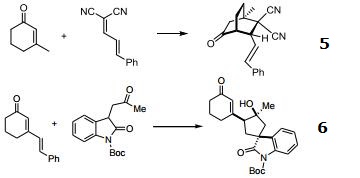
|
| | (8α,9S)-9-aMino-Cinchonan-6'-ol Preparation Products And Raw materials |
|