| Company Name: |
GLP Pharma Standards
|
| Tel: |
+91 9866074638 |
| Email: |
info@glppharmastandards.com |
| Products Intro: |
Product Name:Sulfasalazine EP Impurity-G
CAS:1391062-37-5
Purity:98% Package:50 MG; 100 MG; 250 MG ; 200 MG
|
| Company Name: |
Shenzhen SUNGENING Bio-Medical Co., Ltd.
|
| Tel: |
0755-28967200 13631290199 |
| Email: |
wxf@sungening.com |
| Products Intro: |
Product Name:Sulfasalazine EP Impurity G
Purity:99% HPLC or More Package:10MG;25MG;50MG;100MG
|
|
| | Sulfasalazine EP Impurity G Basic information |
| Product Name: | Sulfasalazine EP Impurity G | | Synonyms: | Sulfasalazine EP Impurity G;XLNQGJSSMSGNMG-ULIFNZDWSA-N;Sulfasalazine Impurity 7(Sulfasalazine EP Impurity G);(3Z)-6-oxo-3-[[4-(pyridin-2-ylsulfamoyl)-2-[4-(pyridin-2-ylsulfamoyl)phenyl]phenyl]hydrazinylidene]cyclohexa-1,4-diene-1-carboxylic acid;Benzoic acid, 5-[2-[4',5-bis[(2-pyridinylamino)sulfonyl][1,1'-biphenyl]-2-yl]diazenyl]-2-hydroxy-;Sulfasalazine N-(Pyridin-2-yl)benzenesulfonamide;5-[2-[4 ,5-bis(pyridin-2-ylsulfamoyl)biphenyl-2-yl]diazenyl]-2-hydroxybenzoic acid;SulfasalazineEPImpurityGDisodiumSalt | | CAS: | 1391062-37-5 | | MF: | C29H22N6O7S2 | | MW: | 630.65 | | EINECS: | | | Product Categories: | | | Mol File: | 1391062-37-5.mol | 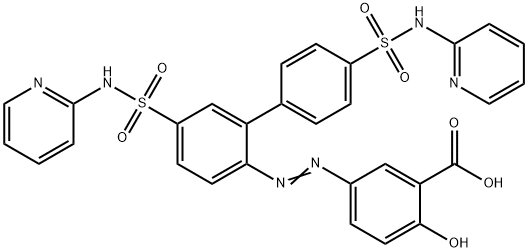 |
| | Sulfasalazine EP Impurity G Chemical Properties |
| Boiling point | 930.0±75.0 °C(Predicted) | | density | 1.51±0.1 g/cm3(Predicted) | | pka | 2.68±0.10(Predicted) |
| | Sulfasalazine EP Impurity G Usage And Synthesis |
| Uses | Sulfasalazine N-(Pyridin-2-yl)benzenesulfonamide is an impurity of Sulfasalazine(S699084). Sulfasalazine is an anti-inflammatory (gastrointestinal). Sulfasalazine has been used in granulomatous colitis. |
| | Sulfasalazine EP Impurity G Preparation Products And Raw materials |
|