- chlorite
-

- $15.99 / 1KG
-
2022-02-25
- CAS:14998-27-7
- Min. Order: 1KG
- Purity: 99%
- Supply Ability: 100KG
|
| | Chlorite Basic information |
| Product Name: | Chlorite | | Synonyms: | Chlorine dioxide ion(1-);Chlorite(1-);Chlorous acid, ion(1-) | | CAS: | 14998-27-7 | | MF: | ClO2 | | MW: | 67.45 | | EINECS: | | | Product Categories: | | | Mol File: | 14998-27-7.mol | 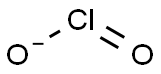 |
| | Chlorite Chemical Properties |
| | Chlorite Usage And Synthesis |
| Definition | An acid known only in solution and as its salts. | | General Description | Crystalline solids or aqueous solutions of these solids. Solids are likely to be water soluble and are denser than water. | | Air & Water Reactions | Probably water soluble. | | Reactivity Profile | Chlorite are oxidizing agents. May liberate oxygen if heated. May react on contact with organic materials such as oil, grease, wood, etc. The reaction may generate sufficient heat to start a fire. Can react with ammonia to give ammonium chlorite, which is shock-sensitive. Mixtures with finely divided metallic or organic substances are highly flammable and may be ignited by friction, Lab. Gov. Chemist(1965). | | Health Hazard | TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Fire may produce irritating and/or toxic gases. Toxic fumes or dust may accumulate in confined areas (basement, tanks, hopper/tank cars, etc.). Runoff from fire control or dilution water may cause pollution. | | Fire Hazard | May explode from friction, heat or contamination. These substances will accelerate burning when involved in a fire. May ignite combustibles (wood, paper, oil, clothing, etc.). Some will react explosively with hydrocarbons (fuels). Containers may explode when heated. Runoff may create fire or explosion hazard. |
| | Chlorite Preparation Products And Raw materials |
|