| Company Name: |
CHEMICAL LAND21
|
| Tel: |
82- 2 -783 - 8063 |
| Email: |
sales21@chemicalland21.com |
| Products Intro: |
|
|
| | piperovatine Basic information |
| Product Name: | piperovatine | | Synonyms: | piperovatine;(2E,4E)-6-(4-Methoxyphenyl)-N-(2-methylpropyl)-2,4-hexadienamide;2,4-Hexadienamide, 6-(4-methoxyphenyl)-N-(2-methylpropyl)-, (2E,4E)- | | CAS: | 25090-18-0 | | MF: | C17H23NO2 | | MW: | 273.37 | | EINECS: | | | Product Categories: | | | Mol File: | 25090-18-0.mol | 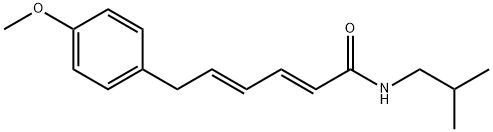 |
| | piperovatine Chemical Properties |
| Melting point | 115-119 °C | | Boiling point | 474.8±37.0 °C(Predicted) | | density | 1.005±0.06 g/cm3(Predicted) | | pka | 14?+-.0.46(Predicted) |
| | piperovatine Usage And Synthesis |
| Description | This alkaloid was first isolated from Piper ovaturn and assigned the empirical
formula C16H2102N, later changed to that given above by Price and Pinder who
also discover the base in Ottonia vehlii Kth. The alkaloid crystallizes from Et20
in colourless needles and has an ultraviolet spectrum containing a single broad
absorption maximum at 262 mil. In the unsaturated side chain, both of the
double bonds are trans. According to Cash, the alkaloid is a temporary depressant
of motor and sensory nerve fibres and of sensory nerve endings. It has the effect
of a heart poison and also as a spinal cord stimulant in frogs bringing about a
tonic spasm somewhat similar to that induced by strychnine. | | References | Dunstan, Garnett., J. Chern. Soc., 67, 94 (1895)
Price, Pinder., J. Org. Chern., 35,2568 (1970)
Pharmacology:
Cash., J. Chern. Soc., 67,94 (1895) |
| | piperovatine Preparation Products And Raw materials |
|