|
|
| | 16,17-Didehydro-9,17-dihydro-9α-hydroxy-12,13-dimethoxyibogamine-18-carboxylic acid methyl ester Basic information |
| Product Name: | 16,17-Didehydro-9,17-dihydro-9α-hydroxy-12,13-dimethoxyibogamine-18-carboxylic acid methyl ester | | Synonyms: | 16,17-Didehydro-9,17-dihydro-9α-hydroxy-12,13-dimethoxyibogamine-18-carboxylic acid methyl ester;Jollyanine;Ibogamine-18-carboxylic acid, 16,17-didehydro-9,17-dihydro-9-hydroxy-12,13-dimethoxy-, methyl ester, (9α)- | | CAS: | 16671-17-3 | | MF: | C23H30N2O5 | | MW: | 414.49 | | EINECS: | | | Product Categories: | | | Mol File: | 16671-17-3.mol | 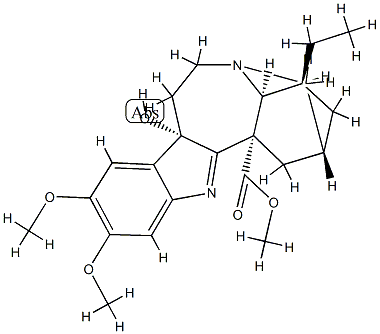 |
| | 16,17-Didehydro-9,17-dihydro-9α-hydroxy-12,13-dimethoxyibogamine-18-carboxylic acid methyl ester Chemical Properties |
| | 16,17-Didehydro-9,17-dihydro-9α-hydroxy-12,13-dimethoxyibogamine-18-carboxylic acid methyl ester Usage And Synthesis |
| Description | Present in Conopharyngia jollyana Stapf., this alkaloid is obtained as colourless
crystals from MeOH and has [α]22D - 56° (c 0.6, CHC13). It contains two
methoxyl groups, one hydroxyl and a C-ethyl group. The ultraviolet spectrum
exhibits absorption maxima at 217,238 and 300 mμ. | | References | Hootele et al., Bull. Soc. Chim. Belg., 76,300 (1967) |
| | 16,17-Didehydro-9,17-dihydro-9α-hydroxy-12,13-dimethoxyibogamine-18-carboxylic acid methyl ester Preparation Products And Raw materials |
|