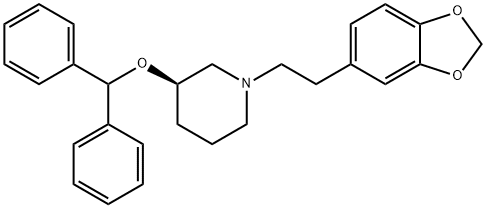
Zamifenacin exhibits moderate oral bioavailability (mouse 26%, rat 64%, dog 100%) and C
max
(mouse 92, rat 905, dog 416 ng/mL) following oral administration (mouse 13.2, rat 20 and, dog 5 mg/kg).
Zamifenacin exhibits terminal elimination half-lives (mouse 2.1, rat 6.0 and, dog 1.1 h) due to high plasma clearance (68, 35, and 39 mL/min/kg respectively) combined with large volumes of distribution (12.5, 19.0, and 3.5 L/kg respectively) following intravenous administration (mouse 5.3, rat 5.0 and, dog 1.0 mg/kg).
|
Animal Model:
|
Male CDl mice (mean weight 23 g)
|
|
Dosage:
|
5.3 mg/kg for i.v.; 13.2 mg/kg for oral (Pharmacokinetic Analysis)
|
|
Administration:
|
Intravenous administration and oral administration
|
|
Result:
|
Oral bioavailability (26%), C
max
(92 ng/mL), T
1/2
(2.1 h).
|
|
Animal Model:
|
Male and female CD rats (mean weight 210 g)
|
|
Dosage:
|
5.0 mg/kg for i.v.; 20 mg/kg for oral (Pharmacokinetic Analysis)
|
|
Administration:
|
Intravenous administration and oral administration
|
|
Result:
|
Oral bioavailability (64%), C
max
(905 ng/mL), T
1/2
(6.0 h).
|
|
Animal Model:
|
Male and two female beagle dogs (13-16 kg)
|
|
Dosage:
|
1.0 mg/kg for i.v.; 5 mg/kg for oral (Pharmacokinetic Analysis)
|
|
Administration:
|
Intravenous administration and oral administration
|
|
Result:
|
Oral bioavailability (100%), C
max
(416 ng/mL), T
1/2
(1.1 h).
|