| Company Name: |
Daicel Chiral Technologies (China)CO.,LTD
|
| Tel: |
021-50460086-9 15921403865 |
| Email: |
han_yajun@dctc.daicel.com |
| Products Intro: |
Product Name:(4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyloxazole
CAS:157904-67-1
Purity:95%/98% Package:100MG;100G; 1KG Remarks:134863
|
(4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyl-Oxazole manufacturers
|
| | (4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyl-Oxazole Basic information |
| Product Name: | (4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyl-Oxazole | | Synonyms: | (4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyl-Oxazole;(4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene 95%;(4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyloxazole],99%e.e.;(4R,4μR,5S,5μS)-2,2μ-(1-Methylethylidene;2,2-Bis[2-(4R,5S-diphenyl-1,3-oxazolinyl)]propane, 95%;(4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyloxazole], 98%, (99% ee);Oxazole, 2,2'-(1-methylethylidene)bis[4,5-dihydro-4,5-diphenyl-, (4R,4'R,5S,5'S)-;(R,S)-BisPh-propabBox | | CAS: | 157904-67-1 | | MF: | C33H30N2O2 | | MW: | 486.6 | | EINECS: | | | Product Categories: | | | Mol File: | 157904-67-1.mol | 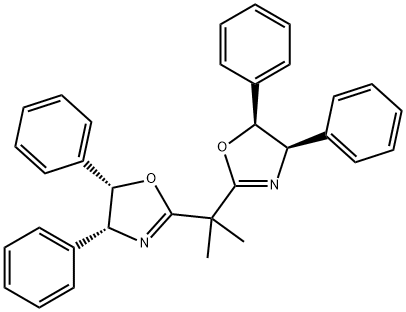 |
| | (4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyl-Oxazole Chemical Properties |
| Melting point | 164-169°C | | Boiling point | 606.7±55.0 °C(Predicted) | | density | 1.16±0.1 g/cm3(Predicted) | | storage temp. | under inert gas (nitrogen or Argon) at 2-8°C | | pka | 4.33±0.70(Predicted) |
| Safety Statements | 24/25 | | WGK Germany | 3 | | HS Code | 29349990 |
| | (4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyl-Oxazole Usage And Synthesis |
| Uses | (4R,4′R,5S,5′S)-2,2′-(1-Methylethylidene) or (4S,5R)-Bis-Phbox) can be used as a ligand:
- To prepare selective exo-catalysts for enantioselective 1,3-dipolar cycloaddition reactions.
- In the asymmetric aminooxygenation of alkenes in the presence of tetramethylaminopyridyl radical (TEMPO) as an oxidant and copper(II) triflate as a catalyst.
- In asymmetric aminofluorination of olefins using an iron catalyst.
This chiral Box ligand was most recently shown to mediate an asymmetric aminofluorination of olefins utlizing Xtalfuor-E (719439) and TREAT-HF (344648). The resulting cyclic carbamates can be readily converted into their concomitant beta-fluoro amino acids. |
| | (4R,4'R,5S,5'S)-2,2'-(1-Methylethylidene)bis[4,5-dihydro-4,5-diphenyl-Oxazole Preparation Products And Raw materials |
|