|
|
| Product Name: | VH-032 | | Synonyms: | VH-032;VHL032;(2S,4R)-1-((S)-2-acetamido-3,3-dimethylbutanoyl)-4-hydroxy-N-(4-(4-methylthiazol-5-yl)benzyl)pyrrolidine-2-carboxamide;L-Prolinamide, N-acetyl-3-methyl-L-valyl-4-hydroxy-N-[[4-(4-methyl-5-thiazolyl)phenyl]methyl]-, (4R)-;(2S,4R)-1-[(2S)-2-acetamido-3,3-dimethylbutanoyl]-4-hydroxy-N-{[4-(4-methyl-1,3-thiazol-5-yl)phenyl]methyl}pyrrolidine-2-carboxamide | | CAS: | 1448188-62-2 | | MF: | C24H32N4O4S | | MW: | 472.6 | | EINECS: | | | Product Categories: | | | Mol File: | 1448188-62-2.mol | 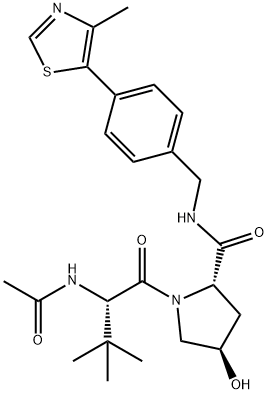 |
| | VH-032 Chemical Properties |
| Boiling point | 778.5±60.0 °C(Predicted) | | density | 1.248±0.06 g/cm3(Predicted) | | solubility | DMSO: 200 mg/mL (423.19 mM) | | pka | 14.07±0.40(Predicted) | | form | Solid | | color | White to off-white |
| | VH-032 Usage And Synthesis |
| Biological Activity | VH032 is a VHL ligand used in the recruitment of the von Hippel-Lindau (VHL) protein. VH032 is a VHL/HIF-1α interaction inhibitor with a Kd
PROTACs contain two different ligands connected by a linker; one is a ligand for an E3 ubiquitin ligase and the other is for the target protein. PROTACs exploit the intracellular ubiquitin-proteasome system to selectively degrade target proteins[1]. | | References | [1]. Michael Zengerle, et al. Selective Small Molecule Induced Degradation of the BET Bromodomain Protein BRD4. ACS Chem Biol. 2015 Aug 21;10(8):1770-7.
[2]. Carles Galdeano, et al. Structure-guided design and optimization of small molecules targeting the protein-protein interaction between the von Hippel-Lindau (VHL) E3 ubiquitin ligase and the hypoxia inducible factor (HIF) alpha subunit with in vitro nanomolar affinities. J Med Chem. 2014 Oct 23;57(20):8657-63.
[3]. Kwok-Ho Chan, et al. Impact of Target Warhead and Linkage Vector on Inducing Protein Degradation: Comparison of Bromodomain and Extra-Terminal (BET) Degraders Derived from Triazolodiazepine (JQ1) and Tetrahydroquinoline (I-BET726) BET Inhibitor Scaffolds. J Med Chem. 2018 Jan 25;61(2):504-513. |
| | VH-032 Preparation Products And Raw materials |
|