- VH-298
-

- $0.00 / 1MG
-
2024-07-22
- CAS:2097381-85-4
- Min. Order: 1MG
- Purity: 98%+ HPLC HNMR
- Supply Ability: 50 g per month
- VH298
-

- $1.00 / 1kg
-
2019-07-06
- CAS:2097381-85-4
- Min. Order: 1kg
- Purity: 95%-99%
- Supply Ability: 100kg
|
| Product Name: | VH298 | | Synonyms: | VH298;VH298;VH 298;CS-2647;(2S,4R)-1-((S)-2-(1-cyanocyclopropanecarboxamido)-3,3-dimethylbutanoyl)-4-hydroxy-N-(4-(4-methylthiazol-5-yl)benzyl)pyrrolidine-2-carboxamide;VH298 USP/EP/BP;(2S,4R)-1-[(2S)-2-[(1-cyanocyclopropanecarbonyl)amino]-3,3-dimethylbutanoyl]-4-hydroxy-N-[[4-(4-methyl-1,3-thiazol-5-yl)phenyl]methyl]pyrrolidine-2-carboxamide;(2S,4R)-1-((S)-2-(1-Cyanocyclopropane-1-carboxamido)-3,3-dimethylbutanoyl)-4-hydroxy-N-(4-(4-methylthiazol-5-yl)benzyl)pyrrolidine-2-carboxamide;E3 ligase-recruiting Moiety,VH-298,Inhibitor,VH298,Ligands for E3 Ligase,VH 298,inhibit | | CAS: | 2097381-85-4 | | MF: | C27H33N5O4S | | MW: | 523.65 | | EINECS: | | | Product Categories: | | | Mol File: | 2097381-85-4.mol | 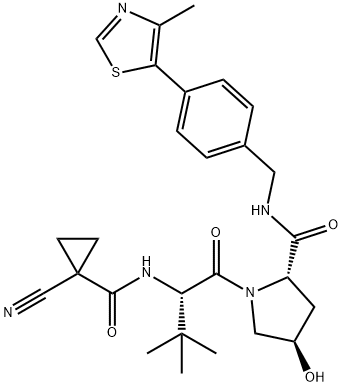 |
| | VH298 Chemical Properties |
| Boiling point | 860.8±65.0 °C(Predicted) | | density | 1.33±0.1 g/cm3(Predicted) | | storage temp. | 2-8°C | | solubility | DMF:30.0(Max Conc. mg/mL);57.29(Max Conc. mM)
DMSO:66.42(Max Conc. mg/mL);126.84(Max Conc. mM)
Ethanol:60.79(Max Conc. mg/mL);116.09(Max Conc. mM)
Ethanol:PBS (pH 7.2) (1:9):0.1(Max Conc. mg/mL);0.19(Max Conc. mM) | | form | A crystalline solid | | pka | 12.28±0.20(Predicted) | | color | Off-white to pink | | InChIKey | NDVQUNZCNAMROD-RZUBCFFCSA-N | | SMILES | C(NCC1=CC=C(C2SC=NC=2C)C=C1)(=O)[C@@H]1C[C@@H](O)CN1C(=O)[C@H](C(C)(C)C)NC(C1(C#N)CC1)=O |
| | VH298 Usage And Synthesis |
| Description | VH298 is a potent VHL (Von Hippel-Lindau, the E3 ligase) inhibitor that stabilizes HIF-α. VH298 blocks the VHL:HIF-α interaction with Kd of 90 nM in isothermal titration calorimetry (ITC) and 80 nM in a competitive fluorescence polarization assay. VH-298 can be used in PROTAC technology. | | In vitro | VH298 can activate the HIF-1 signalling pathway by stabilizing both forms of HIF-1α in vitro. VH298 promotes rFb variability, migration, and synthesis of collagen and regulatory cell factors. VH298 improves the angiogenesis of hUVEC. | | In vivo | VH298 can activate the HIF-1 signalling pathway by stabilizing both forms of HIF-1α in vitro. VH298 promotes rFb variability, migration, and synthesis of collagen and regulatory cell factors. VH298 improves the angiogenesis of hUVEC. | | Description | VH-298 is a E3 ligase ligand-linker conjugate that couples the VHL ligand and blocks the interaction between VHL and HIF-α, initiating hypoxic response. | | Uses | (2S,4R)-1-((S)-2-(1-Cyanocyclopropanecarboxamido)-3,3-dimethylbutanoyl)-4-hydroxy-N-(4-(4-methylthiazol-5-yl)benzyl)pyrrolidine-2-carboxamide is a VHL inhibitors that decreases hypoxia inducible factor-α during hypoxia associated signaling in human cervical cancer, osteosarcoma HeLa and U2OS. cells. | | Biological Activity |
HIF-1α and hydroxy-HIF-1α levels increased in VH298-treated rFb in a time- and dose-dependent manner. Thirty micromolar VH298 could significantly increase cell proliferation, angiogenesis, and gene expression of type I collagen-α1 (Col1-α1), vascular endothelial growth factor A (VEGF-A), and insulin-like growth factor 1 (IGF-1). The VH298-treated wound had a better healing pattern, activation of HIF-1 signaling, and vascularization. It is is a potent VHL inhibitor that stabilizes HIF-α and elicits a hypoxic response via a different mechan: the the blockade of the VHL: HIF-α protein-protein interaction downstream of HIF-α hydroxylation by PHD enzymes. It engages with high affinity and specificity with VHL as its only primary cellular target, leading to selective on-target accumulation of hydroxylated HIF-α in a concentration- and time-dependent fashion in different cell lines, with subsequent upregulation of HIF-target genes at both mRNA and protein levels[1-2].
| | storage | Store at -20°C | | References |
[1] Julianty Frost. “Potent and selective chemical probe of hypoxic signalling downstream of HIF-α hydroxylation via VHL inhibition.” Nature Communications 7 1 (2016).
[2] Shuo Qiu. “Von Hippel-Lindau (VHL) Protein Antagonist VH298 Improves Wound Healing in Streptozotocin-Induced Hyperglycaemic Rats by Activating Hypoxia-Inducible Factor- (HIF-) 1 Signalling.” Journal of Diabetes Research (2019): 1897174.
|
| | VH298 Preparation Products And Raw materials |
|