|
|
| | α- Erythroidine Basic information |
| Product Name: | α- Erythroidine | | Synonyms: | 1,2,6,7-Tetradehydro-12,17-dihydro-3β-methoxy-16(15H)-oxaerythrinan-15-one;a-Erythroidine;1H,12H-Benzo[i]pyrano[3,4-g]indolizin-12-one, 2,6,8,9,9a,10-hexahydro-2-methoxy-, (2R,9aS,13bS)- | | CAS: | 466-80-8 | | MF: | C16H19NO3 | | MW: | 273.33 | | EINECS: | | | Product Categories: | | | Mol File: | 466-80-8.mol | 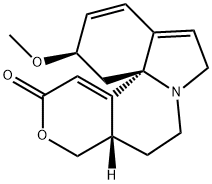 |
| | α- Erythroidine Chemical Properties |
| Melting point | 58-60° | | alpha | D27 +136° (c = 0.5 in water) |
| | α- Erythroidine Usage And Synthesis |
| Purification Methods | Recrystallise -erythroidine from pentane. It is best prepared freshly from the more stable hydrochloride. The hydrochloride (1.3g) in H2O (20mL) is basified with NaHCO3 to pH ~8 and extracted with *C6H6 (6 x 10mL). The combined extracts are evaporated to a small volume and refrigerated. The free base separates as white hygroscopic crystals m 52-55o, which are recrystallised from pentane. Although stable in solution, the crystals turn brown on exposure to air.
-Erythroidine hydrochloride is best purified by dissolving 10g in H2O (100mL), adjusting the pH to 8 with aqueous NaHCO3, extracting with *C6H6 (4 x 20mL), evaporating to 20mL, passing through activated Al2O3 and eluting with *C6H6. The eluate is evaporated to a small volume and the crystals are collected, dissolved in EtOH and dry HCl gas passed through to give the pure hydrochloride. When recrystallised from EtOH, it has m 226-228o(dec), and [ ] D 32 +118o (c 0.5, H2O). It has max 224nm ( 35,500); compare with -erythroidine hydrochloride below. [Boelkeleide & Grundon J Am Chem Soc 75 2563 1953, Boekeleide & Morrison J Am Chem Soc 80 3905 1958, abs config: Hill & Shearer J Org Chem 2 7 3342 1955, Beilstein 27 III/IV 3569.] |
| | α- Erythroidine Preparation Products And Raw materials |
|